Hydrogen Chloride Dalal Simplified ICSE Chemistry Class-10 Solutions. Dalal Simplified ICSE Chemistry Hydrogen Chloride by Dr Viraf and J Dalal for Class 10. Step by step Solutions of Hydrogen Chloride by Dr Viraf and J Dalal of Concept of Simplified Dalal ICSE Chemistry.
Study of Compounds Dalal Simplified ICSE Chemistry Class-10
Get Other Chapter Dalal Simplified ICSE Chemistry Class-10 Solutions
How to Solve Mole Concept ICSE Chemistry Class-10
Note:– Before viewing Solutions of Hydrogen Chloride by Dr Viraf and J Dalal Simplified ICSE Chemistry Solutions .Read the Hydrogen Chloride Dalal Simplified Carefully to understand the concept in better way .After reading the Hydrogen Chloride solve all example of your text book with ICSE Specimen Sample Paper for Class-10 Exam of Council. Focus on Hydrogen Chloride is the Most important Chapter in ICSE Class 10 Chemistry.Previous Year Solved Question Paper for ICSE Board
Additional Questions Hydrogen Chloride Dalal Simplified ICSE
Part – A Hydrogen Chloride
Question 1.
Give a reason why
(a) gastric juices of luammals are acidic
(b) HCl is considered a polar covalent compound.
Answer:
(a) HCI gas occurs in free state in gastric juices of mammals.
(b) Due to difference in electronegativities of H and Cl ; The bond in HCl is a polar covalent.
Question 2.
Give the equation for preparation of HCI gas by Synthesis. State two conditions involved in the synthesis.
Answer:
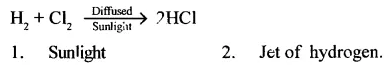
Question 3.
Give a balanced equation for preparation of HCl gas in the laboratory from sodium chloride.
Answer:
![]()
Question 4.
In the laboratory preparation of HCl frem sodium chloride, state why the following are
preferred –
- Cone. H2SO4 as a reactant
- Temp, below 200°C
- Cone. H2SO4 as a drying agent.
Answer:
- Since it is non-volatile and has a high boiling point.
- The glass apparatus does not crack, no hard crust is formed, fuel is not wasted.
- It only removes moisture content of the gas but not react with it.
Question 5.
State with reasons the method of collection of HCl gas in the laboratory.
Answer:
HCl gas is collected by upward displacement of air because it is 1.28 times heavier than air. It is not collected in water because it is highly soluble in water.
Question 6.
Compare the density of HCl gas with air and state the solubility of HCl gas in water.
Answer:
V.D of HCl = 18.25, V.D of air = 14.4. It is highly soluble in water.
Question 7.
State why HCl gas forms a mist of droplets of HCl acid in moist air.
Answer:
Due to high solubility, HCl gas fumes in moist air and forms a mist of droplets of HCl acid.
Question 8.
State what the fountain experiment demonstrates with reference to HCl gas.
Answer:
Fountain experiment demonstrates the solubility of HCI gas in water and its acidic nature.
Question 9.
State the colour change in three different indicators in presence of HCl gas.
Answer:
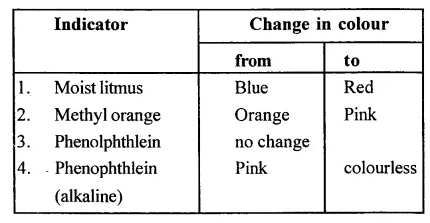
Question 10.
Give a balanced equation for the thermal dissociation of
- a gas
- a solid (both containing the chloride ion).
Answer:
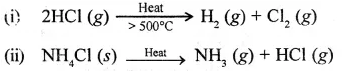
Question 11.
Give the equation and state the observation seen when HCl gas reacts with ammonia.
Answer:
When a gas jar containing hydrogen chloride gas is inverted over ajar full of ammonia gas, dense white fumes are formed.
![]()
Question 12.
Convert iron to iron (II) chloride using HCl gas.
Answer:
![]()
Question 13.
Explain the arrangement (i) not used (ii) used – for converting HCl gas into HCl acid.
Answer:
Hydrochloric acid is prepared by dissolving hydrogen chloride gas in water usi ig a special funnel arrangement.
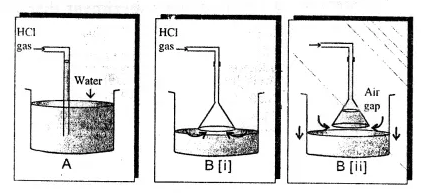
1. Direct absorption of HCl gas in water using a delivery tube i.e., arrangement (i) causes back suction.
Hydrogen chloride gas is extremely soluble in water. If a delivery tube through which HCl gas is passed is directly immersed in water, the rate of absorption of HCl gas is high and hence a partial vacuum is created in the tube. The pressure outside being higher causes the water to be pushed up into the delivery tube and damages the apparatus. This is called back-suction.
2. Special funnel arrangement, i.e., arrangement (ii) is used for avoiding back suction.
The funnel arrangement :
(a) Prevents or minimizes back-suction of water.
(b) Provides a large surface area for absorption of HCl gas.
The rim of the funnel is placed so that it just touches the trough containing water. If back-suction occurs, the water rises up the funnel and the level outside the funnel falls, thus creating an air gap between the rim of the funnel and the surface of water. The pressure outside and inside equalize and the water which had risen in the funnel falls down again. This process continues till the water in the trough is saturated with hydrogen chloride gas resulting information of hydrochloric acid.
Hence hydrochloric acid is not prepared in the laboratory by passing hydrogen chloride gas directly through water, but prepared using a special funnel arrangement.
Question 14.
Explain the term ‘constant boiling mixture’.
Answer:
A solution of HCl in water forms a constant boiling mixture with water.
Question 15.
State why dilute HCl cannot be concentrated beyond a certain concentration by boiling.
Answer:
On boiling the mixture evolves out the vapours of both acid and water in the same proportion as in the liquid. Hence dil HCl cannot be concentrated beyond a certain concentration.
Question 16.
Name the ions obtained when HCl dissociates in aqueous solution.
Answer:
Hydronium ions.
Question 17.
Name the ion responsible for acidic nature of HCl acid.
Answer:
The presence of hydrogen ions |H+| imparts acidic nature to HCl.
Question 18.
State which of two – a solution of HCl in water or in toluene is an electrolyte, giving reasons.
Answer:
A solution of hydrogen chloride in water; water being a covalent solvent ionizes aqueous solution of HCl shows acidic properties and is an electrolyte, whereas toluene is not; being not a polar solvent.
Question 19.
Give four different word equations relating to acidic properties of an aq. soln. of HCl gas.
Answer:
Zinc + Hydrochloric acid → Zinc chloride + Hydrogen
Sodium hydroxide + Hydrochloric acid → Sodium chloride + Water
Sodium carbonate + Hydrochloric acid → Sodium chloride + Carbon dioxide + Water
Calcium oxide + Hydrochloric acid → Calcium chloride + Water
Question 20.
Give balanced equations to obtain
- H2
- CO2
- SO2
- H2S from dil. HCl.
Answer:
- Zn + 2HCl → ZnCl2 + H2
- Na2CO2 + 2HCl → 2NaCl + H2O + CO2
- Na2SO3 + 2HCl → 2NaCl + H20 + SO2
- FeS + 2HCl → FeCl2 + H2S
Question 21.
Convert two soluble metallic nitrates to insoluble metallic chlorides using dil. HCl.
Answer:

Question 22.
State how you would prove that HCl contains
- hydrogen – using an active metal below magnesium
- chlorine – using an oxidising agent not containing lead.
Answer:
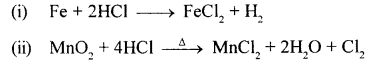
Question 23.
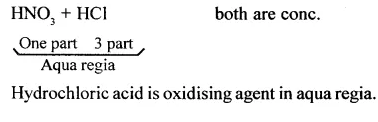
State the composition of aqua regia. State which component is the oxidising agent in aqua regia.
Answer:
Question 24.
Convert hydrochloric acid to nascent chlorine.
Answer:
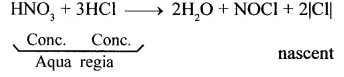
Question 25.
State why aqua regia dissolves gold, which is insoluble in all other acids.
Answer:
In aqua regia (a mixture of 1 part cone. HNO3 and 3 parts cone. HCl by volume), nitric acid oxidises hydrochloric acid to give nascent chlorine. This nascent chlorine is very reactive. It reacts with gold to give gold (III) chloride, which is soluble in water.
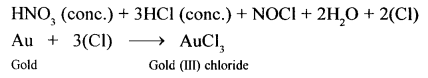
Question 26.
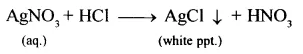
Give three tests for hydrochloric acid. Convert silver nitrate to a soluble salt of silver using hydrochloric acid and an alkali.
Answer:
- If glass rod dipped in ammonia soln. (NH4OH) brought near vapours of hydrochloric acid.
Dense white fumes of – ammonium chloride are formed. - Addition of silver nitrate soln. to dil. HCl (acidified with dil. HNO3)
Curdly white precipitate – of silver chloride obtained. - Action of heat on a mixture of manganes dioxide and cone. HCl Greenish yellow gas (chlorine).
Question 27.
State two industrial products manufactured from hydrochloric acid, which are also manufactured from nitric and sulphuric acid. Give two general uses of hydrochloric acid.
Answer:
Industrial products manufactured from HCl acid are : Dyes, paints, drugs.
General uses of hydrochloric acid are :
- Hydrochloric acid dissolves the calcium phosphate present in bones.
- Hydrochloric acid dissolves the metallic oxides coating on the surface of metals. Thereby cleans the metallic surface.
–Try Also :–


Thanks for Help 🙌☺️
ok and keep in touch