Calorimetry: Concept of Heat, Law of Calorimetry ,Specific Heat, Latent Heat. Application of High Specific Heat of Water. Application of High Latent Heat of Water. Concept of Calorimetry. Numericals on Calorimetry. Deference between Heat and Temperature.
Calorimetry:- Chapter 11 Concise Physics 10th Solution
Exercise 11 A
Question 1
Define the term heat.
Answer 1
The kinetic energy due to random motion of the molecules of a substance is known as its heat energy.
Question 2
Name the S.I. unit of heat.
Answer 2
joule (J)
Question 3
Define the term calorie. How is it releated to joule?
Answer 3
One calorie of heat is the heat energy required to raise the temperature of 1 g of water from 14.5oC to 15.5 oC.
1 calorie = 4.186 J
Question 4
Define one kilo calorie of heat.
Answer 4
One kilo-calorie of heat is the heat energy required to raise the temperature of 1 kg of water from 14.5oC to 15.5oC.
Question 5
Define temperature and name its S.I. unit.
Answer 5
The quantity which determines the direction of flow of heat between two bodies kept in contact is called temperature.
S.I. unit kelvin (K).
Question 6
State three differences between heat and temperature.
Answer 6
| Heat | Temperature |
| The kinetic energy due to random motion of the molecules ofa substance is known as its heat energy. | The quantity which determines the direction of flow of heat between two bodies kept in contact is called temperature. |
| S.I. unit joule (J). | S.I. unit kelvin (K). |
| It is measured by the principle of calorimetry. | It is measured by a thermometer. |
Question 7
Define calorimetry.
Answer 7
The measurement of the quantity of heat is called calorimetry.
Question 8
Define the term heat capacity and state its S.I. unit.
Answer 8
The heat capacity of a body is the amount of heat energy required to raise its temperature by 1oC or 1K.
S.I. unit is joule per kelvin (JK-1).
Question 9
Define the term specific heat capacity and state its S.I. unit.
Answer 9
The specific heat capacity of a substance is the amount of heat energy required to raise the temperature of unit mass of that substance through by 1oC (or 1K).
S.I. unit is joule per kilogram per kelvin (J kg-1K-1).
Question 10
How is heat capacity of a body releated to specific heat capacity of its substance?
Answer 10
Heat capacity = Mass x specific heat capacity
Question 11
State three differences between the heat capacity and specific heat capacity.
Answer 11
Heat capacity of the body is the amount of heat required to raise the temperature of (whole) body by 1whereas specific heat capacity is the amount of heat required to raise the temperature of unit mass of the body by 1.
Heat capacity of a substance depends upon the material and mass of the body. Specific heat capacity of a substance does not depend on the mass of the body.
S.I. unit of heat capacity is and S.I. unit of specific heat capacity is.
Question 12
Name a liquid which has the highest specific heat capacity.
Answer 12
Water has the highest specific heat capacity.
Question 13
Write the approximate value of specific heat capacity of water in S.I. unit.
Answer 13
Specific heat capacity of water=4200 J kg-1 K-1
Question 14
What do u mean by the following statement;
(i) The heat capacity of a body is 50 J/k ?
(ii) The specific heat capacity of copper is 0.4 J/g/K ?
Answer 14
(i) The heat capacity of a body is 50JK-1 means to increase the temperature of this body by 1K we have to supply 50 joules of energy.
(ii) The specific heat capacity of copper is 0.4Jg-1K-1 means to increase the temperature of one gram of copper by 1K we have to supply 0.4 joules of energy.
Question 15
Specific heat capacity of a substance A is 3.8 J g-1 K-1 and of substance B is 0.4 Jg-1 k-1. Which substance is a good conductor of heat? How did you arrive at your conclusion?
Answer 15
The specific heat capacity of substance B is lesser than that of A. So, for same mass and same heat energy, the rise in temperature for B will be more than that of A. Hence, substance B is a good conductor of heat.
Question 16
Name two factors on which the heat energy librated by a body on cooling depends.
Answer 16
Change in temperature and the nature of material
Question 17
Name three factors on which the heat energy absorbed by a body depends and state how does it depend on them
Answer 17
The quantity of heat energy absorbed by a body depends on three factors :
(i)Mass of the body – The amount of heat energy required is directly proportional to the mass of the substance.
(ii)Nature of material of the body – The amount of heat energy required depends on the nature on the substance and it is expressed in terms of its specific heat capacity c.
(iii)Rise in temperature of the body – The amount of heat energy required is directly proportional to the rise in temperature.
Question 18
Write the expression for the heat energy Q received by m kg of a substance of specific heat capacity c J kg-1 K-1 when it is heated through  toC.
toC.
Answer 18
The expression for the heat energy Q
Q= mct (in joule)
Question 19
Same amount of heat is supplied to two liquid A and B. The liquid A shows a greater rise in temperature. What can you say about the heat capacity of A as compared to that of B?
Answer 19
Heat capacity of liquid A is less than that of B.
As the substance with low heat capacity shows greater rise in temperature.
Two blocks P and Q of different metals having their mass in the ratio 2:1 are given same amount of heat. Their temperatures rise by same amount, compare their specific heat capacities
Answer 20

Question 21
What is the principle of method of mixture? What other name is given to it? Name the law on which this principle is based.
Answer 21
The principle of method of mixture:
Heat energy lost by the hot body = Heat energy gained by the cold body.
This principle is based on law of conservation of energy.
Question 22
A mass m1 of a substance of specific heat capacity c1 at temperature T1 is mixed with a mass m2 of other substance of specific heat capacity c2 at a lower temperature T2. Deduce the expression for the temperature of mixture. State assumption made, if any.
Answer 22
A mass m1 of a substance A of specific heat capacity c1 at temperature T1 is mixed with a mass m2 of other substance B of specific heat capacity c2 at a lower temperature T2 and final temperature of the mixture becomes T.
Fall in temperature of substance A = T1 – T
Rise in temperature of substance B = T – T2
Heat energy lost by A = m1 c1 fall in temperature
= m1c1(T1 – T)
Heat energy gained by B= m2 c2 rise in temperature
= m2c2(T – T2)
If no energy lost in the surrounding, then by the principle of mixtures,
Heat energy lost by A = Heat energy gained by B
m1c1(T1 – T)= m2c2(T – T2)
After rearranging this equation, we get
Here we have assumed that there is no loss of heat energy.
Question 23
Why do the farmers fill their fields with water on a cold winter night?
Answer 23
In the absence of water, if on a cold winter night the atmospheric temperature falls below 0oC, the water in the fine capillaries of plant will freeze, so the veins will burst due to the increase in the volume of water on freezing. As a result, plants will die and the crop will be destroyed. In order to save the crop on such cold nights, farmers fill their fields with water because water has high specific heat capacity, so it does not allow the temperature in the surrounding area of plants to fall up to 0oC.
Question 24
Discuss the role of high specific heat capacity of water with reference to climate in coastal areas
Answer 24
The specific heat capacity of water is very high. It is about five times as high as that of sand. Hence the heat energy required for the same rise in temperature by a certain mass of water will be nearly five times than that required by the same mass of sand. Similarly, a certain mass of water will give out nearly five times more heat energy than that given by sand of the same mass for the same fall in temperature. As such, sand gets heated or cooled more rapidly as compared to water under the similar conditions. Thus a large difference in temperature is developed between the land and the sea due to which land and sea breezes are formed. These breezes make the climate near the sea shore moderate.
Question 25
The reason is that water does not cool quickly due to its large specific heat capacity, so a hot water bottle provides heat energy for fomentation for a long time.
Question 26
Water property of water makes it an effective coolant?
Answer 26
By allowing water to flow in pipes around the heated parts of a machine, heat energy from such part is removed. Water in pipes extracts more heat from surrounding without much rise in its temperature because of its large specific heat capacity. So, Water is used as an effective coolant.
Question 27
Give one example each where high specific heat capacity of water is used (i) as coolant, (ii) as heat reservoir
Answer 27
(i)Radiator in car.
(ii)To avoid freezing of wine and juice bottles
Question 28
A liquid X has specific heat capacity higher than the liquid Y. Which liquid is useful as (i) coolant in car radiators and, (ii) heat reservoir to keep juice bottles without freezing?
Answer 28
The specific heat capacity of liquid X is higher than that of Y. So, for same mass and same heat energy, the rise in temperature for X will be less than that of Y.
(i) As a coolant in car radiators, the liquid needs to absorb more energy without much change in temperature. So, liquid X is ideal for this function.
(ii) As a heat reservoir to keep juice bottles without freezing, the liquid needs to give out large amount of heat before reaching freezing temperatures. Hence, liquid X is ideal for this function.
Question 29
(a) What is calorimeter?
(b)Name the material of which it is made of. Give two reasons for using the material stated by you.
(c) Out of the three metals A, B and C of specific heat 900 J kg-1 °C-1, 380 J kg-1 °C-1 and 460 J kg-1 °C-1 respectively, which will you prefer for calorimeter? Given reason.
(d) How is the loss of heat due to radiation minimized in a calorimeter?
Answer 29
(a)A calorimeter is a cylindrical vessel which is used to measure the amount of heat gained or lost by a body when it is mixed with other body.
(b)It is made up of thin copper sheet because:
(i) Copper is a good conductor of heat, so the vessel soon acquires the temperature of its contents.
(ii)Copper has low specific heat capacity so the heat capacity of calorimeter is low and the amount of heat energy taken by the calorimeter from its contents to acquire the temperature of its contents is negligible.
(c)Heat capacity of the calorimeter should be low so out of three metals the one which have lowet specific heat capacity should be preferred.
(d) By polishing the outer and inner surface of the vessel the loss due to radiation can be minimized.
Question 30
Why the base of a cooking pan is made thick and heavy?
Answer 30
By making the base of a cooking pan thick, its thermal capacity becomes large and it imparts sufficient heat energy at a low temperature to the food for its proper cooking. Further it keeps the food warm for a long time, after cooking.
MULTIPLE CHOICE TYPE (Exercise 11A)
1. The S.I. unit of heat capacity is :
(a) J kg-1
(b) J K-1
(c) J kg-1 K-1
(d) cal 0C-1
Answer 1
![]()
Question 2
2. The S.I. unit of specific heat capacity is :
(a) J kg-1
(b) J K-1
(c) J kg-1 K-1
(d) kilocal kg-10C-1
Answer 2
J kg-1 K-1
Question 3
3. The specific heat capacity of water is :
(a) 4200 J kg-1 K-1
(b) 420 J g-1 K-1
(c) 0.42 J g-1 K-1
(d) 4.2 J kg-1 K-1
Answer 3
4200 J kg-1 K-1
ICSE Class 10th Calorimetry Numerical (Chapter 11 A)
Question 1
By imparting heat to a body, its temperature rises by 150C. What is the corresponding rise in temperature on the Kelvin scale?
Answer 1.
The size of 1 degree on the Kelvin scale is the same as the size of 1 degree on the Celsius scale. Thus, the difference (or change) in temperature is the same on both the Celsius and Kelvin scales.
Therefore, the corresponding rise in temperature on the Kelvin scale will be 15K.
Question 2.
a)Calculate the heat capacity of a copper vessel of mass 150 g if the specific heat capacity of copper is 410 J kg-1 K-1.
(b)How much heat energy will be required to increase the temperature of the vessel in part (a) from 25oC to 35oC?
Answer 2
(i) Mass of copper vessel=150 g
=0.15 kg
The specific heat capacity of copper = 410 J kg-1 K-1.
Heat capacity= Mass X specific heat capacity
=0.15 kg X 410Jkg-1K-1
=61.5JK-1
Change in temperature= (35-25)oC=10oC=10K
(ii) Energy required to increase the temperature of vessel
=0.15 X 410 X 10
=615 J
Question 3
A piece of iron of mass 2.0 kg has a thermal capacity of 966 J K-1. Find
(i) Heat energy needed to warm it by 15oC, and
(ii) Its specific heat capacity in S.I unit.
Answer 3
(i)We know that heat energy needed to raise the temperature by 15o is = heat capacity x change in temperature.
Heat energy required= 966 J K-1 x 15 K = 14490 J.
(ii)We know that specific heat capacity is = heat capacity/ mass of substance
So specific heat capacity is = 966 / 2=483 J kg-1 K-1.
Question 4
Calculate the amount of heat energy required to raise the temperature of 100 g of copper from 20oC to 70oC. Specific heat of capacity of copper =390 J kg-1 K-1.
Answer 4
Mass of copper m = 100 g = 0.1 kg
Change of temperature t = (70-20)oC
Specific heat of capacity of copper =390 J kg-1 K-1
Amount of heat required to raise the temperature of 0.1 kg of copper is
Q =
= 0.1 x 50 x 390
= 1950 J
Question 5
1300 J of heat energy is supplied to raise the temperature of 0.5 kg of lead from 20oC to 40oC. Calculate the specific heat capacity of lead.
Answer 5
Heat energy supplied = 1300 J
Mass of lead = 0.5 kg
Change in temperature = (40-20)oC = 20 oC (or 20 K)
Specific heat capacity of lead
c = 130 J kg-1 K-1
Question 6
Find the time taken by a 500 W heater to raise the temperature of 50 kg of material of specific heat capacity 960 J kg-1 K-1, from 18oC to 38oC. Assume that all the heat energy supplied by the heater is given to the material.
Answer 6
Specific heat capacity of material c =960 J kg-1 K-1
Change in temperature T=(38-18)oC = 20oC (or 20 K)
Power of heater P = 500 W
Time taken by a heater to raise the temperature of material
t= 1920 seconds
t=32 min
Question 7
An electric heater of power 600 W raises the temperature of 4.0 kg of a liquid from 10oC to 15oC in 100 s. calculate :
(i)the specific heat capacity of the liquid.
(ii)the heat capacity of 4.0 kg of liquid.
Answer 7
Power of heater P= 600 W
Mass of liquid m=4.0 kg
Change in temperature of liquid = (15-10)oC = 5oC(or 5 K)
Time taken to raise its temperature =100s
Heat energy required to heat the liquid
![]()
and
![]() =600X100=60000J
=600X100=60000J
![]() =
=![]()
Heat capacity= c x m
Heat capacity= 4 x 3000JKg-1 K-1 = 1.2 x 104 J/K
Question 8
0.5 kg of lemon squash at 30oC is placed in a refrigerator which can remove heat at an average rate of 30 J s-1. How long will it take to cool the lemon squash to 5oC? Specific heat capacity of squash = 4200 J kg-1 K-1.
Answer 8
Change in temperature= 30 – 5 = 25 K.
![]()
![]()
![]() =
=![]()
t=29 min 10 sec.
Question 9
A mass of 50g of a certain metal at 150° C is immersed in 100g of water at 11° C. The final temperature is 20° C. Calculate the specific heat capacity of the metal. Assume that the specific heat capacity of water is 4.2 J g-1 K-1.
Solution 9
Heat liberated by metal=![]()
Heat absorbed by water=![]()
Heat energy lost= heat energy gained
=![]() =
=![]()
S=0.52 J g-1 K-1.
Question 10
45 g of water at 50oC in a beaker is cooled when 50 g of copper at 18oC is added to it. The contents are stirred till a final constant temperature is reached. Calculate this final temperature. The specific heat capacity of copper is 0.39 Jg-1 K-1 and that of water is 4.2 J g-1 K-1.
Answer 10
Mass of water (m1)=45 g
Temperature of water (T1) =50oC
Mass of copper (m2) =50 g
temperature of copper(T2) =18oC
Final temperature (T) =?
The specific heat capacity of the copper c2 = 0.39 J/g/K
The specific heat capacity of water c1 = 4.2 J/g/K
![]()
![]()
![]() oC
oC
T= 47oC.
Question 11
200g of hot water at 80oC is added to 300 g of cold water at 10oC. Neglecting the heat taken by the container, calculate the final temperature of the mixture of the water. Specific heat capacity of water = 4200 J kg-1 K-1.
Answer 11
Mass of hot water (m1) = 200g
Temperature of hot water (T1) = 80oC
Mass of cold water (m2) = 300g
Temperature of cold water (T2) =10oC
Final temperature (T) =?
![]() c1=c2
c1=c2
![]()
![]()
T=38oC.
Question 12
The temperature of 600 g of cold water rises by 15oC when 300 g of hot water at 50oC added to it. What was the initial temperature of the cold water
Answer 12
Mass of hot water (m1) = 300 g
Temperature (T1) = 50oC
Mass of cold water (m2) = 600 g
Change in temperature of cold water (T-T2) =15oC
Final temperature =ToC
The specific heat capacity of water is c.
![]()
![]()
T = 20oC.
Final temperature = 20o c
Change in temperature = 15oC
Initial temperature of cold water = 20oC -15oC = 5oC.
Question 13
1.0 kg of water is contained in a 1.25 kW kettle. Calculate the time taken for the temperature of water to rise from 25°C to its boiling point 100°C. Specific heat capacity of water = 4.2 J g-1 K-1.
Answer 13
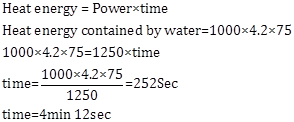
EXERCISE- (11) B
Question 1
(a)What do you understand by the change of phase of substance?
(b)Is there any change in temperature during the change of phase?
(c)Does the substance absorb or liberate any heat during the change of phase?
(d) what is the name given to the energy absorbed during a phase change?
Answer 1
(a) The process of change from one state to another at a constant temperature is called the change of phase of substance.
(b)There is no change in temperature during the change of phase.
(c)Yes, the substance absorbs or liberates heat during the change of phase.
(d) Latent heat
Question 2
A substance changes from its solid state to the liquid state when heat is supplied to it
(a) Name the process.
(b) What name is given to heat observed by the substance.
(c) How does the average kinetic energy of the molecules of the substance change.
Answer 3
(a) Melting.
(b) Latent heat of melting.
(c) As their id no change in temperature the average kinetic energy of the molecules does not change.
Question 3
A substance undergoes (i) a rise in its temperature, (ii) A change in its phase without change in its temperature. In each case, state the energy change in the molecules of the substance.
Answer 3
(i)Average kinetic energy of molecules changes.
(ii)Average potential energy of molecules changes.
Question 4
How does the (a) average kinetic energy (b) average potential energy of molecules of a substance change during its change in phase at a constant temperature, on heating?
Answer 4
(a) Average kinetic energy does not change.
(b) Average potential energy increases.
Explanation: When a substance is heated at constant temperature (i.e. during its phase change state), the heat supplied makes the vibrating molecules gain potential energy to overcome the intermolecular force of attraction and move about freely. This means that the substance changes its form.
However, this heat does not increase the kinetic energy of the molecules, and hence, no rise in temperature takes place during the change in phase of a substance.
This heat supplied to the substance is known as latent heat and is utilized in changing the state of matter without any rise in temperature.
Question 5
State the effect of presence of impurity on the melting point of ice. Give one use of it.
Answer 5
The melting point of ice decreases by the presence of impurity in it.
Use: In making the freezing mixture by adding salt to ice. This freezing mixture is used in preparation of ice creams.
Question 7
The diagram shows the change of phases of a substance on temperature time graph.
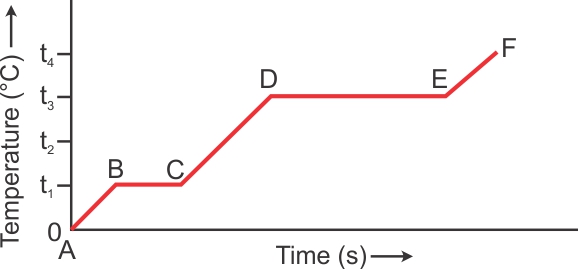
(a)What do parts AB, BC, CD and DE represent?
(b) What is the melting point of the substance?
(c) What is the boiling point of the substance?
Answer 7
(a) AB part shows rise in temperature of solid from 0 to T1oC, BC part shows melting at temperature T1oC, CD part shows rise in temperature of liquid from T1oC to T3oC , DE part shows the boiling at temperature T3oC.
(b) T1oC.
(c) T3oC.
Question 8
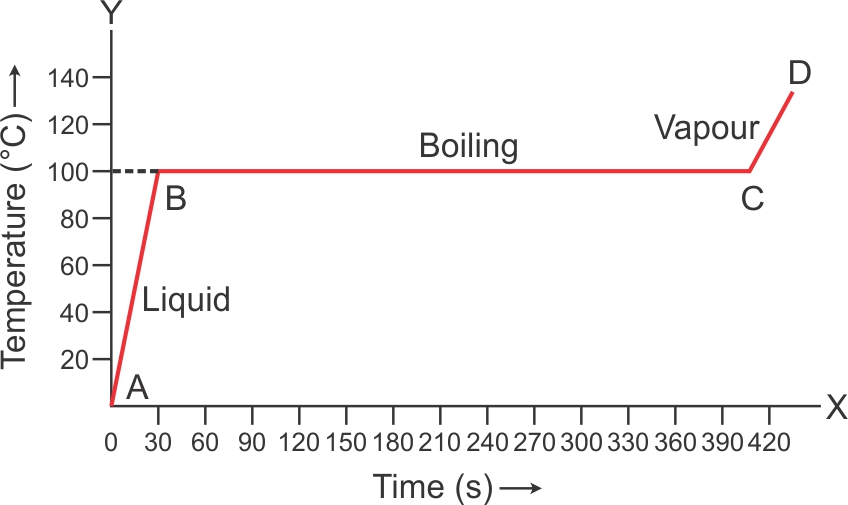
1 kg of ice at 0o is heated at constant rate and its temperature is recorded after every 30 s till steam is formed at 100o C. Draw a temperature time graph to represent the change of phase
Answer 8
Question 9
Explain the terms boiling and boiling point.
Answer 9
Boiling: The change from liquid to gaseous phase on heating at a constant temperature is called boiling.
Boiling point: The particular temperature at which vaporization occurs is called the boiling point of liquid.
Question 10
How is the volume of water affected when it boils at 100oC?
Answer 10
Volume of water wills increases when it boils at 100oC.
Question 11
How is the boiling point of water affected when some salt is added to it?
Answer 11
The boiling point of water increases on adding salt.
Question 12
What is the effect of increase in pressure on the boiling point of a liquid?
Answer 12
The boiling point of a liquid increases on increasing the pressure.
Question 13
Water boils at 120 °C in a pressure cooker. Explain the reason
Answer 13
- The boiling point of a liquid increases with the increase in pressure and decreases with the decrease in pressure.
- The boiling point of pure water at one atmospheric pressure (= 760 mm of Hg) is 100 °C.
- In a pressure cooker, the water boils at about 120 °C to 125 °C due to increase in pressure, as the steam is not allowed to escape out of it.
Question 14
Write down the approximate range of temperature at which the water boils in a pressure cooke
Answer 14
In a pressure cooker, the water boils at about 120oC to 125oC.
Question 15
It is difficult to cook vegetables on hills and mountains. Explains the reason.
Answer 15
This is because at high altitudes atmospheric pressure is low; therefore boiling point of water decreases and so it does not provide the required heat energy for cooking.
Question 16
Complete the following sentences:
(a)When ice melts, its volume………….
(b)Decrease in pressure over ice ………….. its melting point.
(c)Increase in pressure ………..the boiling point of water.
(d)A pressure cooker is based on the principle that boiling point of water increases with
……………….
(e)The boiling point of water is defined as ………………………..
(f) water can be made to boil at 115°C by …………….. pressure over its surface.
Answer 16
(a) When ice melts, its volume decreases.
(b) Decrease in pressure over ice increases its melting point.
(c) Increase in pressure increases the boiling point of water.
(d)A pressure cooker is based on the principle that boiling point of water increases with the increase in pressure.
(e) The boiling point of water is defined as the constant temperature at which water changes to steam.
(f) water can be made to boil at 115°C by increasing pressure over its surface.
Question 17
What do you understand by the term latent heat?
Answer 17
Latent heat: The heat energy exchanged in change of phase is not externally manifested by any rise or fall in temperature, it is considered to be hidden in the substance and is called the latent heat.
Question 18
Define the term specific latent heat of fusion of ice. State its S.I. unit.
Answer 18
The quantity of heat required to convert unit mass of ice into liquid water at (melting point) is called the specific latent heat of fusion of ice.
Its S.I. unit is Jkg-1.
Question 19
Write the approximate value of specific latent heat of ice.
Answer 19
Specific latent heat of ice: 336000 J kg-1.
Question 20
‘The specific latent heat of fusion of ice is 336 J g-1 ‘. Explain the meaning of this statement.
Answer 20
It means 1 g of ice at 0oC absorbs 360 J of heat energy to convert into water at 0oC.
Question 21
1 g ice at 0o C melts to form 1 g water at 0o. State whether the latent heat is absorbed or given out by ice.
Answer 21
Latent heat is absorbed by ice.
Question 22
Which has more heat: 1 g of ice at 0o C or 1 g of water at 0oC? Give reasons.
Answer 22
1 g of water at 0oC has more heat than 1 g of ice at 0oC. This is because ice at 0oC absorbs 360 J of heat energy to convert into water at 0oC.
Question 23
(a) Which requires more heat: 1 g ice at 0o C or 1 g water at 0oC to raise its temperature to 10oC? (b) Explain your answer in part (a).
Answer 23
(a) 1 g ice at 0oC requires more heat because ice would require additional heat energy equal to latent heat of melting.
(b) 1 g ice at 0oC first absorbs 336 J heat to convert into 1 g water at 0oC.
Question 24
Ice cream appears colder to the mouth than water at 0oC. Give reasons.
Answer 24
This is because 1 g of ice at 0oC takes 336 J of heat energy from the mouth to melt at 0oC. Thus mouth loses an additional 336 J of heat energy for 1 g of ice at 0oC than for 1g of water at 0oC. Therefore cooling produced by 1 g of ice at 0oC is more than for 1g of water at 0oC.
Question 25
The soft drink bottles are cooled by (i) ice cubes at 0°C, and (ii) iced-water at 0°C. Which will cool the drink quickly? Give reason
Answer 25
This is because 1 g of ice at 0oC takes 336 J of heat energy from the bottle to melt into water at 0oC. Thus bottle loses an additional 336 J of heat energy for 1 g of ice at 0oC than for 1 g iced water at 0oC. Therefore bottled soft drinks get cooled, more quickly by the ice cubes than by iced water.
Question 26
It is generally cold after a hailed storm than during and before the hail storm. Give reasons.
Answer 26
The reason is that after the hail storm, the ice absorbs the heat energy required for melting from the surrounding, so the temperature of the surrounding falls further down and we feel colder
Question 27
The temperature of surroundings starts falling when ice in a frozen lake starts melting. Give reasons.
Answer 27
The reason is that the heat energy required for melting the frozen lake is absorbed from the surrounding atmosphere. As a result, the temperature of the surrounding falls and it became very cold.
Question 28
Water in lakes and ponds do not freeze at once in cold countries. Give reason
Answer 28
The specific latent heat of fusion of ice is sufficiently high, about 336 J g-1. Before freezing the water in the lakes and ponds will have to release a large quantity of heat to the surrounding. If there is any layer of ice formed on water than water being a poor conductor of heat will also prevent the loss of heat from water of lake. Hence, in cold countries water in lakes and ponds do not freeze.
Question 29
Explain the following:
(a) The surroundings become pleasantly warm when water in a lake starts freezing in cold countries.
(b) The heat supplied to a substance during its change of state, does not cause any rise in its temperature
Answer 29
(a) The reason is that the specific latent heat of fusion of ice is sufficiently high, so when the water of lake freezes, a large quantity of heat has to be released and hence the surrounding temperature becomes pleasantly warm.
(b) Heat supplied to a substance during its change of state, does not cause any rise in its temperature because this is latent heat of phase change which is required to change the phase only.
Multiple Choice Type (Chapter 11 B)
Question 1
The S.I. unit of specific latent heat is :
(a) cal g-1
(b) cal g-1 K-1
(c) J kg-1
(d) J kg -1 K-1
Answer 1
J kg-1
Question 2
The specific latent heat of fusion of water is :
(a) 80 cal g-1
(b) 2260 J g-1
(c) 80 J g-1
(d) 336 J kg-1
Answer 2
80cal g-1
Numerical Problem (Exercise 11 B) Calorimetry
Question 1
10g of ice at 0oC absorbs 5460J of heat energy to melt and change to water at 50oC. Calculate the specific latent heat of fusion of ice. Specific heat capacity of water is 4200Jkg-1K-1.
Answer 1
Mass of ice=10g = 0.01kg
Amount of heat energy absorbed, Q=5460J
Specific latent heat of fusion of ice=?
Specific heat capacity of water = 4200Jkg-1K-1
Amount of heat energy required by 10g (0.01kg) of water at 0oC to raise its temperature by 50oC= 0.01X4200X50=2100J.
Let Specific latent heat of fusion of ice=L Jg-1.
Then,
Q = mL + mcT
5460 J =10 x L + 2100J
L=336Jg-1.
Question 2
How much heat energy is released when 5.0 g of water at 20oC changes into ice at 0oC? Take specific heat capacity of water =4.2 J g-1 K-1, specific latent heat of fusion of ice =336 J g-1.
Answer 2
Mass of water m = 5.0 g
specific heat capacity of water c = 4.2 J g-1 K-1
specific latent heat of fusion of iceL =336 J g-1
Amount of heat energy released when 5.0 g of water at 20oC changes into water at 0oC = 5 x 4.2 x 20 = 420 J.
Amount of heat energy released when 5.0g of water at 0oC changes into ice at 0oC=5x336J=1680J.
Total amount of heat released =1680 J + 420 J = 2100 J.
Question 3
A molten metal of mass 150 g is kept at its melting point 800oC. When it is allowed to freeze at the same temperature, it gives out 75000 J of heat energy.
(a)What is the specific latent heat of the metal?
(b) If the specific heat capacity of metal is 200 J kg-1 K-1, how much additional heat energy will the metal give out in cooling to -50oC?
Answer 3
Mass of metal =150 g
Specific latent heat of metal
![]()
Specific heat capacity of metal is 200 J kg-1 K-1.
Change in temperature= 800-(-50) = 850oC (or 850 K).
![]()
Question 4
A refrigerator converts 100g of water at 20oC to ice at -10oC in 73.5 min. calculate the average rate of heat extraction in watt. The specific heat capacity of water is 4.2 Jg-1K-1, specific latent heat of ice is 336Jg-1 and the specific heat capacity of ice is 2.1Jg-1K-1.
Answer 4
Amount of heat released when 100g of water cools from 20o to 0oC =100X20X4.2=8400J.
Amount of heat released when 100g of water converts into ice at 0oC =100X336=33600J.
Amount of heat released when 100g of ice cools from 0oC to -10oC =100X10X2.1=2100J.
Total amount of heat=8400+33600+2100=44100J.
Time taken= 73.5min=4410s.
Average rate of heat extraction (power)
![]()
Question 5
In an experiment, 17 g of ice is used to bring down the temperature of 40 g of water at 34oC to its freezing temperature. The specific heat capacity of water is 4.2 J g-1 K-1. Calculate the specific latent heat of ice. State one important assumption made in the above calculation.
Answer 5
Mass of ice m1 =17 g
Mass of water m2 =40 g.
Change in temperature =34-0=34K
Specific heat capacity of water is 4.2Jg-1K-1.
Assuming there is no loss of heat, heat energy gained by ice (latent heat of ice), Q= heat energy released by water
Q = 40 x 34 x 4.2 = 5712 J.
Specific latent heat of ice=.![]()
Question 6
Find the result of mixing 10g of ice at -10oC with 10g of water at 10oC. Specific heat capacity of ice is 2.1Jg-1K-1, specific latent heat of ice is 336Jg-1, and specific heat capacity of water is 4.2Jg-1 K-1.
Answer 6
Let whole of the ice melts and let the final temperature of the mixture be ToC.
Amount of heat energy gained by 10g of ice at -10oC to raise its temperature to 0oC= 10x10x2.1=210J
Amount of heat energy gained by 10g of ice at 0oC to convert into water at 0oC=10×336=3360 J
Amount of heat energy gained by 10g of water (obtained from ice) at 0oC to raise its temperature to ToC = 10×4.2x(T-0)=42T
Amount of heat energy released by 10g of water at 10oC to lower its temperature to ToC = 10×4.2x(10-T)=420-42T
Heat energy gained = Heat energy lost
210 + 3360 + 42T = 420-42T
T = -37.5oC
This cannot be true because water cannot exist at this temperature.
So whole of the ice does not melt. Let m gm of ice melts. The final temperature of the mixture becomes 0oC.
So, amount of heat energy gained by 10g of ice at -10oC to raise its temperature to 0oC= 10x10x2.1=210J
Amount of heat energy gained by m gm of ice at 0oC to convert into water at 0oC=mx336=336m J
Amount of heat energy released by 10g of water at 10oC to lower its temperature to 0oC = 10×4.2x(10-0)=420
Heat energy gained = Heat energy lost
210 + 336m = 420
m = 0.625 gm
Question 7
A piece of ice of mass 40 g is added to 200 g of water at 50oC. Calculate the final temperature of water when all the ice has melted. Specific heat capacity of water is 4200 J kg-1 K-1, specific latent heat of fusion of ice =336 x 103 J kg-1.
Answer 7
Let final temperature of water when all the ice has melted =ToC.
Amount of heat lost when 200g of water at 50oC cools to ToC=
200X4.2X(50-T) = 42000-840T
Amount of heat gained when 40g of ice at 0oC converts into water at 0oC.= 40X336J = 13440 J
Amount of heat gained when temperature of 40g of water at 0oC rises to ToC= 40X4.2X(T-0) = 168T
We know that
Amount of heat gained=amount of heat energy lost.
13440+168T= 42000-840T
168T+840T= 42000-13440
1008T= 28560
T=28560/1008=28.33oC.
Question 8
250 g of water at 30o C is contained in a copper vessel of mass 50 g. Calculate the mass of ice required to bring down the temperature of vessel and its contents to 5o C. specific latent heat of fusion of ice = 336 x 103 J kg-1, specific heat capacity of copper = 400 J kg-1 K-1, specific heat capacity of water is 4200 J kg-1 K-1.
Answer 8
Mass of copper vessel m1 = 50 g.
Mass of water contained in copper vessel m2 = 250 g.
Mass of ice required to bring down the temperature of vessel = m
Final temperature = 5o C.
Amount of heat gained when ‘m’ g of ice at 0o C converts into water at 0o C = m × 336 J
Amount of heat gained when temperature of ‘m’ g of water at 0o C rises to 5o C = m × 4.2 × 5
Total amount of heat gained = m × 336 + m × 4.2 × 5
Amount of heat lost when 250 g of water at 30o C cools to 5o C =
250 × 4.2 x 25 = 26250 J
Amount of heat lost when 50 g of vessel at 30o C cools to 5o C =
50 x 0.4 × 25 = 500 J
Total amount of heat lost = 26250 + 500 = 26750 J
We know that amount of heat gained = amount of heat lost
m × 336 + m × 4.2 × 5 = 26750
357 m = 26750
m = 26750/357 = 74.93 g
Hence, mass of ice required is 74.93 g.
Question 9
2 kg of ice melts when water at 100oC is poured in a hole drilled in a block of ice. What mass of water was used? Specific heat capacity of water is 4200 J kg-1 K-1, specific latent heat of fusion of ice = 336 J g-1.
Answer 9
Since the whole block does not melt and only 2 kg of it melts, so the final temperature would be 0 oC.
Amount of heat energy gained by 2 kg of ice at 0oC to convert into water at 0oC=2X336000=672000 J
Let amount of water poured=m kg.
Initial temperature of water =100oC.
Final temperature of water =0oC.
Amount of heat energy lost by m kg of water at 100oC to reach temperature 0oC =mX4200X100 = 420000m J
We know that heat energy gained =heat energy lost.
672000J= mX420000J
m=672000/420000=1.6kg
Question 10
Calculate the total amount of heat energy required to convert 100 g of ice at -10o C completely into water at 100o C. Specific heat capacity of ice 2.1 J g-1 K-1, specific heat capacity of water is 4.2 J g-1 K-1, specific latent heat of fusion of ice = 336 J g-1
Answer 10
Amount of heat energy gained by 100 g of ice at -10o C to raise its temperature to 0o C =
100 × 2.1 × 10 = 2100 J
Amount of heat energy gained by 100 g of ice at 0o C to convert into water at 0o C =
100 × 336 = 33600 J
Amount of heat energy gained when temperature of 100 g of water at 0o C rises to 100o C =
100 × 4.2 × 100 = 42000 J
Total amount of heat energy gained is = 2100 + 33600 + 42000 = 77700 J = 7.77 × 104 J
Question 11
The amount of heat energy required to convert 1 kg of ice at -10oC completely into water at 100oC is 777000 J. calculate the specific latent heat of ice. Specific heat capacity of ice = 2100 J kg-1 K-1, Specific heat capacity of water is 4200 J kg-1 K-1.
Answer 11
Amount of heat energy gained by 1kg of ice at -10oC to raise its temperature to 0oC= 1 x 2100 x 10 = 21000 J
Amount of heat energy gained by 1kg of ice at 0oC to convert into water at 0oC=L
Amount of heat energy gained when temperature of 1kg of water at 0oC rises to 100oC= 1 x 4200 x 100 = 420000 J
Total amount of heat energy gained = 21000+420000+L=441000 +L.
Given that total amount of heat gained is =777000J.
So,
441000+L=777000.
L=777000-441000.
L=336000JKg-1
Question 12
200 g of ice at 0 °C converts into water at 0 °C in 1 minute when heat is supplied to it at a constant rate. In how much time, 200 g of water at 0 °C will change to 20 °C? Take specific latent heat of ice = 336 J g-1.
Answer 12
Mass of ice, mice = 200 g
Time for ice to melt, t1 = 1 min = 60 s
Mass of water, mw = 200 g
Temperature change of water, ΔT = 20 °C
Rate of heat exchange is constant. So, power required for converting ice to water is same as the power required to increase the temperature of water.
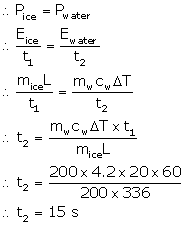
Hence, the time required is 15 seconds.
Get Other Chapter Class 10th Concise Physics Solution
- Chapter 1 – Force
- Chapter 2 – Work, Energy and Power
- Chapter 3 – Machines
- Chapter 4 – Refraction of Light at Plane Surfaces
- Chapter 5 – Refraction through Lens
- Chapter 7 – Sound
- Chapter 8 – Current Electricity
- Chapter 9 – Household Circuits
- Chapter 10 – Electro-Magnetism
- Chapter 11- Calorimetry
Class 10th Concise Biology Solution
Concise Biology ICSE Class 10 Solution Chapter Wise
Class 10th ML Aggrwal Maths Solution
ML Aggarwal 10th Maths Solution Chapterwise