Chemical Bonding ICSE Class-10 Concise Chemistry Selina Solutions Chapter-2. We Provide Step by Step Answer of Exercise-2, with Intex -1, Intex-2 and Previous Year Questions of Exercise-2 Chemical Bonding ICSE Class-10 . Visit official Website CISCE for detail information about ICSE Board Class-10.
Chemical Bonding ICSE Class-10 Concise Chemistry Selina Solutions Chapter-2
| Board | ICSE |
| Publications | Selina Publishers PVT LTD |
| Subject | Concise Chemistry |
| Class | 10th |
| writer | Dr SP Singh |
| Chapter-2 | Chemical Bonding |
| Topics | Intext -1 & 2 and Exercise-2 Questions Solutions |
| Edition | 2021-2022 |
-: Select Topics :-
Intext 1 ,
Intext 2 ,
Concise Chemistry (Selina Publication ) Solution Intext 1 Chapter-2 Chemical Bonding
Page-25
Question 1
How do atoms attain noble gas configuration?
Answer 1
Atoms lose, gain or share electrons to attain noble gas configuration.
Question 2
Define
(a) a chemical bond
(b) an electrovalent bond
(c) a covalent bond
Answer 2
(a) A chemical bond may be defined as the force of attraction between any two atoms, in a molecule, to maintain stability.
(b) The chemical bond formed between two atoms by transfer of one or more electrons from the atom of a metallic electropositive element to an atom of a non-metallic electronegative element is called as electrovalent bond.
(c) The chemical bond formed due to mutual sharing of electrons between the given pairs of atoms of non-metallic elements is called as a covalent bond.
Question 3
What are the conditions for formation of an electrovalent bond?
Answer 3
Conditions for formation of Ionic bond are:
(i) The atom which changes into cation should possess 1, 2 or 3 valency electrons. The other atom which changes into anion should possess 5, 6 or 7 electrons in the valence shell.
(ii) A high difference of electronegativity of the two atoms is necessary for the formation of an Ionic bond.
(iii) There must be an overall decrease in energy i.e., energy must be released.
For this an atom should have low value of Ionisation potential and the other atom should have high value of electron affinity.
(iv) Higher the lattice energy, greater will be the case of forming an ionic compound.
Question 4
An atom X has three electrons more than noble gas configuration. What type of ion will it form? Write the formula of its (i) Sulphate (ii) Nitrate (iii) Phosphate (iv) carbonate (v) Hydroxide
Answer 4
It will form a cation: X3+
(i) X2(SO4)3
(ii) X(NO3)3
(iii) XPO4
(iv) X2(CO3)3
(v) X(OH)3
Question 5
Mention the basic tendency of an atom which makes it combine with other atoms.
Answer 5
Atoms combine with other atoms to attain stable octet or noble gas configuration.
Question 6
The element X has the electronic configuration 2, 8, 18, 8, 1. Without identifying x,
(a) predict the sigh and charge on a simple ion of X.
(B) write if X be an oxidizing agent or reducing agent and why.
Answer 6
(a) X+
(b) X will be a strong reducing agent as it will have the tendency to donate its valence electron.
Question 7
In the formation of compound XY2, an atom X gives one electron to each Y atom, what is the nature of bond in XY2? Draw the electron dot structure of this compound?
Answer 7
X and Y form an ionic bond in XY2.
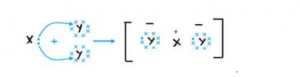
Question 8
An atom has 2, 8, 7 electrons in its shell. It combines with Y having 1 electron in its outermost shell.
(a) What type of bond will be formed between X and Y?
(b) Write the formula of compound formed.
Answer 8
(a) X has 7 electrons in its outermost shell and Y has only one electron in its outermost shell so Y loses its one electron and X gains that electron to form an ionic bond.
(b) The formula of the compound would be XY.
Question 9
explain with the help of ionic equations Draw electron dot structural diagrams the formation of following electrovalent compound
(i) NaCl (ii) MgCl2 (iii) CaO.
Answer 9
(i) Orbit structure and electron dot diagram of NaCl:
Sodium chloride is a compound formed from the ionic bonding of sodium and chloride. Sodium chloride is formed when sodium atoms interact with chlorine atoms. Sodium will donate an electron (which is a negatively-charged particle) to chlorine. This makes sodium slightly positive and chlorine slightly negative. Sodium becomes Na+ and Chlorine becomes Cl- Thus sodium is reduced in this process as it donates the electron and chlorine gain electron and is oxidized.

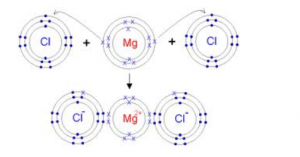
(ii )Orbit structure and electron dot diagram of MgCl2:
The formation of magnesium chloride can be thought of as a reaction involving magnesium metal, Mg, and chlorine gas, Cl2
- A magnesium atom, which is a metal in Group II A, tends to lose its 2 outer-shell valence electrons to become a magnesium ion, (i.e. cation). The magnesium metal is said to be oxidized.
- The covalently bonded Cl2 molecule gains two electrons to become two chloride ions, (i.e. anions). Chlorine is said to be reduced. The two electrons lost by the magnesium is gained by the chlorine atom and thus produce magnesium ion and two chloride ions.
- The oppositely charged of the magnesium and chloride ions attract each other and ionic bonds are formed. Each cation is surrounded by anions, and each anion is surrounded by cations. The simplest formula for this ionic compound is MgCl2.
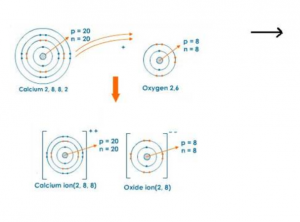
(iii)Electron dot Orbit structure diagram of CaO:
The Calcium ion is an Alkaline earth metal and wants to give up the 2 s orbital elections and become a +2 cation.
Oxygen has six valence electrons and is looking to gain two electrons to complete the octet (8) electron count in the valence shell making it a -2 anion.
When the charges of the Calcium +2 and the Oxygen -2 are equal and opposite, the ions for an electrical attraction. (Remember Paula Abdul told us “Opposites Attract”)
This one to one ratio of charges makes the formula CaO.
Question 10
Compare :
(a) Sodium atom and sodium ion
(b) Chlorine atom and chlorine ion
With respect to
(i) Atomic structure
(ii) Electrical state
(iii) Chemical action and
(iv) toxicity
Answer 10
(a) Sodium atom and sodium ion
(i) Sodium atom has one electron in M shell while sodium ion has 8 electrons in L shell.
(ii) Sodium atom is neutral while sodium ion is positively charged.
(iii) Sodium atom is highly reactive while its ion is inert.
(iv)Sodium atom is poisonous while sodium ion is non-poisonous.
(b)Chlorine atom and chlorine ion
(i) Chlorine atom has 7 electrons in its M shell while Chloride ion has 8 electrons in the same shell.
(ii) Chlorine atom is neutral while chloride ion is negatively charged.
(iii) Chlorine atom is highly reactive while its ion is inert.
(iv)Chlorine gas is poisonous while chloride ion is non-poisonous.
Question 11
The electronic configuration of fluoride ion is the same as that of a neon atom. What is the difference between two?
Answer 11
Fluoride ion is negatively charged while neon atom is neutral.
Question 12
State Which of the following is reduction reaction and Which is oxidation .
(i) Pb → Pb2+ + 2e–
(ii) Fe2+ – e– → Fe3+
(iii) A3+ + e-1 → A2+
(iv) Cu → Cu2+
Answer 12
(i) Pb → Pb2+ + 2e– = Oxidation Reaction
(ii) Fe2+ – e– → Fe3+ = Oxidation Reaction
(iii) A3+ + e-1 → A2+ = Reduction Reaction
(iv) Cu → Cu2+ = Reduction Reaction
Page-26
Question-13
What do you understand by redox reactions? Explain oxidation and reduction in terms of loss or gain of electrons.
Answer-13
Transfer of electron(s) is involved in the formation of an electrovalent bond. The electropositive atom undergoes oxidation, while the electronegative atom undergoes reduction. This is known as a redox process.
Oxidation: In the electronic concept, oxidation is a process in which an atom or ion loses electron(s).
Zn → Zn2+ + 2e–
Reduction: In the electronic concept, the reduction is a process in which an atom or ion accepts electron(s).
Cu2+ + 2e–→ Cu
Question-14
Divide the following redox reactions into oxidation and reduction half reactions.
(i) Zn + Pb2+ → Zn2+ + Pb
(ii) Zn + Cu2+ → Zn2+ + Cu
(iii) Cl2 + 2Br– → Br2 + 2Cl–
(iv) Sn2+ + 2Hg2+ → Sn4+ + Hg2+
(v) 2Cu+ → Cu + Cu2+
Answer-14
i. Zn→ Zn2+ + 2e– (Oxidation)
Pb2+ + 2e– → Pb (Reduction)
ii. Zn→ Zn2+ + 2e– (Oxidation)
Cu2+ + 2e–→ Cu (Reduction)
iii. Cl2 + 2e–→ 2Cl– (Reduction)
2Br–→ Br2 + 2e– (Oxidation)
iv. Sn2+→ Sn4+ + 2e– (Oxidation)
2Hg2+ + 2e–→ Hg2 (Reduction)
v. Cu+→ Cu2+ + e– (Oxidation)
Cu+ + e– → Cu (Reduction)
Question-15
Potassium (Atomic No. 19) and chlorine (Atomic No. 17) react to form a compound. On the basis of electronic concept, explain
(i) oxidation
(ii) reduction
(iii)oxidising agent
(iv)reducing agent
Answer-15
Potassium (Atomic No. 19) and chlorine (Atomic No. 17) react to form a compound. On the basis of electronic concept, explain
(i) oxidation
(ii) reduction
(iii)oxidising agent
(iv)reducing agent
ICSE Chemistry Class-10 Concise Selina Solution Chemical Bonding Chapter 2 Intex 2
Page-32
Question 1
What are conditions necessary for the formation of covalent molecules?
Answer 1
(i) Both atoms should have four or more electrons in their outermost shells, i.e., non-metals.
(ii) Both the atoms should have high electronegativity.
(iii) Both the atoms should have high electron affinity and high ionisation potential.
(iv) Electronegativity difference between the two atoms should be zero or negligible.
(v) The approach of the atoms towards one another should be accompanied by decrease of energy.
Question 2
Elements A, B and C have atomic numbers 17, 19 and 10 respectively.
(a) State which one is:
(i) A non-metal
(ii) A metal
(iii) Chemically inert?
(b) Write down the formula of the compound formed by two of the above elements.
Answer 2
(a) A is a non-metal; B is a metal while C is a chemically inert element.
(b) BA
Question 3
Draw the electron dot diagram and structure of:
(a) nitrogen molecule
(b) magnesium chloride
(c) methane (2017)
Answer 3
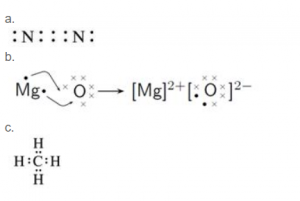
Question 4
What is the difference between:
(a) Ionic compounds and polar covalent compounds
(b) Ionic compounds and covalent compounds
(c) A polar covalent compound and a non-polar covalent compound?
Answer 4
(a) difference between Ionic compounds and polar covalent compounds
Ionic compounds are formed as a result of the transfer of one or more electrons from the atom of a metallic electropositive element to an atom of a non-metallic electronegative element.
A polar covalent compound is the one in which there is an unequal distribution of electrons between the two atoms.
(b) difference between Ionic compounds and covalent compounds
Ionic compounds, made up of ions, are generally crystalline solids with high melting and boiling points.
They are soluble in water and good conductors of electricity in aqueous solution and molten state.
Covalent compounds, made up of molecules, can exist as soft solids or liquids or gases with low melting and boiling points. They are generally insoluble in water and poor conductors of electricity.
(c) difference between Ionic Polar covalent and Non-polar compounds
Polar covalent compounds are formed between 2 non-metal atoms that have different electronegativities and therefore have unequal sharing of the bonded electron pair.
Non-polar compounds are formed when two identical non-metals equally share electrons between them.
Question 5
A solid is crystalline, has a high melting point and is water soluble. Describe the nature of the solid.
Answer 5
The crystalline solid is ionic in nature. It has strong electrostatic forces of attraction between its ions, which cannot be separated easily.
Crystalline solids have high melting and boiling points, and a large amount of energy is required to break the strong bonding force between ions.
Water is a polar compound, so it decreases the electrostatic forces of attraction in the crystalline solid, resulting in free ions in the aqueous solution. Hence, the solid dissolves.
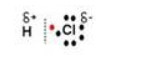
Question 6
What do you understand by polar covalent compounds? Explain it by taking hydrogen chloride as an example.
Answer 6
Covalent compounds are said to be polar when shared pair of electrons are unequally distributed between the two atoms. For example in HCl, the high electronegativity of the chlorine atom attracts the shared electron pair towards itself. As a result, it develops a slight negative charge and hydrogen atom develops a slight positive charge. Hence, a polar covalent bond is formed.
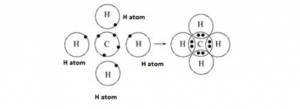
Question 7
(a) Explain the bonding in methane molecule using the electron dot structure. (2015)
(b) The methane molecule is a non-polar molecule. Explain.
Answer 7
(a)
| Atom | Electronic configuration | Nearest noble gas | To attain stable electronic configuration of a nearest noble gas |
| Carbon | 126C [2,4] | Neon [2,8] | Carbon needs four electrons to complete the octet. |
| Hydrogen | 11H [1] | Helium [2] | Hydrogen needs one electron to complete the duplet. |
One atom of carbon shares four electron pairs, one with each of the four atoms of hydrogen.
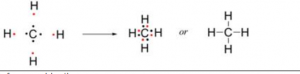
| Before combination
(4 [H] and 1 [C] atom) |
After combination (CH4 – Methane molecule)
|
(b) Methane is a covalent compound and is non-polar in nature. This is because the shared pair of electrons is equally distributed between the two atoms. So, no charge separation takes place and the molecule is symmetrical and electrically neutral.
Question 8
Give the characteristic properties of:
(a) Electrovalent compounds
(b) Covalent compounds
Answer 8
(a) Properties of Electrovalent Compounds:
- Ionic compounds usually exist in the form of crystalline solids.
- They have high melting and boiling points.
- These compounds are generally soluble in water but insoluble in organic solvents.
- They are good conductors of electricity in the fused or in an aqueous solution state.
(b) Properties of Covalent Compounds:
1.The covalent compounds exist as gases or liquids or soft solids.
- The melting and boiling points of covalent compounds are generally low.
- Covalent compounds are insoluble in water but dissolve in organic solvents.
- They are non-conductors of electricity in the solid, molten or aqueous state.
Question 9
(a) State the type of bond is formed when the combining atoms have:
(i) zero E.N. difference
(ii) small E.N. difference
(iii) large E.N. difference
- State the type of bond formed, and draw Lewis structure of
- water
- calcium oxide
Answer 9
(a)
(i) Covalent bond
(ii) Polar covalent bond
(iii). Ionic bond
(b)
(i) water: Polar covalent bonding takes place in water.
![]()
(ii) calcium oxide: Electrovalent bonding takes place in calcium oxide.
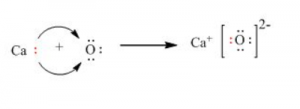
Question 10
Explain the following:
(a) Electrovalent compounds conduct electricity.
(b) Electrovalent compounds have a high melting point and boiling point, while covalent compounds have low melting and boiling points.
(c) Electrovalent compounds dissolve in water, whereas covalent compounds do not.
(d) Electrovalent compounds are usually hard crystals yet brittle.
(e) Polar covalent compounds conduct electricity.
(f) Water is a polar covalent molecule ?
Answer 10
(a) Electrovalent compounds
are good conductors of electricity in the fused or aqueous state because electrostatic forces of attraction between ions in the solid state are very strong and these forces weaken in the fused state or in the solution state. Hence, ions become mobile.
(b) Electrovalent compounds
have a strong force of attraction between the oppositely charged ions, so a large amount of energy is required to break the strong bonding force between ions. So, they have high boiling and melting points.
Covalent compounds have weak forces of attraction between the binding molecules, thus less energy is required to break the force of binding. So, they have low boiling and melting points.
(c) As water is a polar compound, it decreases the electrostatic forces of attraction, resulting in free ions in the aqueous solution. Hence, electrovalent compounds dissolve.
Covalent compounds do not dissolve in water but dissolve in organic solvents. Organic solvents are non-polar; hence, these dissolve in non-polar covalent compounds.
(d) Electrovalent compounds
are usually hard crystals yet brittle because they have strong electrostatic forces of attraction between their ions which cannot be separated easily.
(e) Polar covalent compounds conduct electricity because they form ions in their solutions.
(f) This means that in the water molecule, one side is positively charged and the other side is negatively charged. Since the water molecule (comprising two atoms of Hydrogen and one of Oxygen) is formed by covalent bonds, the electrons are shared.
This sharing causes the electrons to stay closer to the oxygen atom giving it a slight negative charge while the hydrogen atoms, as a consequence, have a slight positive charge.
Question 11
Elements X, Y and Z have atomic numbers 6, 9 and 12, respectively. Which one
(a) forms an anion
(b) forms a cation
(c) State the type of bond between Y and Z and give its molecular formula.
Answer 11
(a)
(i) Y = 9
(ii) Z = 12
(b) Ionic bond with molecular formula ZY2.
Question 12
Taking MgCl2 as an electrovalent compound and CCl4 as a covalent compound, give four differences between electrovalent and covalent compounds.
Answer 12
| MgCl2 – Electrovalent compound | CCl4 – Covalent compound |
| hard crystalline solids consisting of ions. | gases or liquids or soft solids. |
| high melting and boiling points. | low melting and boiling points. |
| conduct electricity in the fused or aqueous state. | do not conduct electricity in the solid, molten or aqueous state. |
| It is soluble in inorganic solvents but insoluble in organic solvents. | It is insoluble in water but dissolve in organic solvents. |
Question 13
Potassium chloride is an electrovalent compound, while hydrogen chloride is a covalent compound. But both conduct electricity in their aqueous solutions. Explain.
Answer 13
Potassium chloride is an electrovalent compound and conducts electricity in the molten or aqueous state because the electrostatic forces of attraction weaken in the fused state or in aqueous solution.
Polar covalent compounds like hydrogen chloride ionise in their solutions and can act as an electrolyte. So, both can conduct electricity in their aqueous solutions.
Question 14
(a) Draw the electron dot structure of covalent compound methane (non polar) and HCL (polar) and give two difference between them .
(b) Name two compounds that are covalent when pure but produce ions when dissolved in water.
(c) For each compound mentioned above, give the formulae of ions formed in the aqueous solution.
Answer 14
(a)
Non-polar covalent compound (Methane,CH4)
1. Covalent compounds are said to be non-polar when shared pair of electrons are equally distributed between the two atoms.
2. No charge separation takes place. The covalent molecule is symmetrical and electrically neutral.
Polar covalent compound (HCl)
1. Covalent compounds are said to be polar when shared pair of electrons are unequally distributed between the two atoms. 2. Charge separation takes place. The atom which attracts electrons more strongly develops a slight negative charge while the other develops a slight positive charge
(b) HCl and NH3
(c) HCl + H2O → H3O+ + Cl–
NH3 + H2O →NH4+ + OH–
Question 15
An element M burns in oxygen to form an ionic bond MO. Write the formula of the compounds formed if this element is made to combine with chlorine and sulphur separately.
Answer 15
Formula of compound when combined with sulphur – MSFormula of compound when combined with chlorine – MCl2
Question 16
Element A has 2 electrons in its M shell. Element B has atomic number 7.
(a) Write equations to show how A and B form ions.
(b) If B is a diatomic gas, write the equation for the direct combination of A and B to form a compound.
(c) If the compound formed between A and B is melted and an electric current is passed through the molten compound, then element A will be obtained at the _________ and B at the ________ of the electrolytic cell.
Answer 16
(c) If the compound formed between A and B is melted and an electric current is passed through the molten compound, then element A will be obtained at the cathode and B at the anode of the electrolytic cell.
Chapter-2 Chemical Bonding Selina Solution for ICSE Chemistry Exercise-2
page-35
Question 1
Define a coordinate bond and give conditions for its formation.
Answer 1
The bond formed between two atoms by sharing a pair of electrons, provided entirely by one of the combining atoms but shared by both is called a coordinate bond. It is represented by an arrow starting from the donor atoms and ending in the acceptor atom.
Conditions:
- One of the two atoms must have at least one lone pair of electrons.
- Another atom should be short of at least a lone pair of electrons.
The two lone pair of electrons in the oxygen atom of water is used to form coordinate bond with the hydrogen ion which is short of an electron resulting in the formation of the hydronium ion.
H2O + H+ H3O+ Over here the hydrogen ion accepts one lone pair of electrons of the oxygen atom of water molecule leading to the formation of a coordinate covalent bond.
Question 2
What do you understand by lone pair of electron? (2018)
Answer 2
A pair of electrons which is not shared with any other atom is known as a lone pair of electrons. It is provided to the other atom for the formation of a coordinate bond.
Question 3
State the type of bonding in the following molecules:
(a) Water
(b) Calcium oxide
(c) Hydroxyl ion
(d) Methane
(e) Ammonium ion
(f) Ammonium chloride
Answer 3
(a) Polar covalent bond
(b) Ionic bond
(c) O and H are bonded with a single covalent bond and oxygen possesses a single negative charge in the hydroxyl ion.
(d) Covalent bond
(e) Coordinate bond
(f) Electrovalent bond, dative bond (or coordinate bond) and covalent bond
Question 4
(a) Draw an electron dot diagram to show the structure of each of the following:
(i) Hydronium ions (2018)
(ii) Ammonium ion
(iii) Hydroxyl ion
State the type of bonding present in them.
(b) Give two examples in each case:
(i) Co-ordinate bond compounds
(ii) Solid covalent compounds
(iii) Gaseous polar compounds
(iv) Gaseous non-polar compounds
(v) Liquid non-polar compounds
Answer 4
(a)
(i)
(ii) 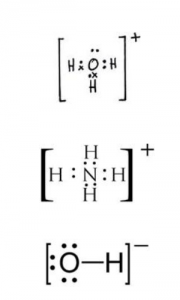
(iii)
(b)
(i) Ammoniumion and hydronium ion
(ii) Phosphoruspentachloride and diamond
(iii) Hydrogen chloride and water vapour
(iv) Oxygen gas and nitrogen gas
(v) Toluene and Gasoline
Question 5
Element M forms a chloride with the formula MCl2 which is a solid with high melting point. M would most likely be in the group in which ______ is placed. [(a) Na (b) Mg (c)Al (d) Si]
Answer 5
Mg
Question 6
Complete the following:
| Sodium | Phosphorus | Carbon | |
| Formula of chloride | |||
| Nature of bonding | |||
| Physical state of chloride |
Answer-6
| Sodium | Phosphorus | Carbon | |
| Formula of chloride | NaCl | PCl5 | CCl4 |
| Nature of bonding | Ionic | Covalent | Covalent |
| Physical state of chloride | Solid | Solid | Liquid |
Questions-7
(a) How many atoms of each kind are present in the following molecules: calcium oxide, chlorine, water, carbon tetrachloride?
(b) How many electrons are required by each atom mentioned in (a) to attain the nearest noble gas configuration?
Answer-7
(a) CaO- 1 calcium atom + 1 oxygen atom
Cl2 – 2 chlorine atoms
H2O – 2 hydrogen atoms + 1 oxygen atom
CCl4 – 1 carbon atom + 4 chlorine atoms
(b) Ca – will donate two electrons
O – will accept two electrons
Cl – will accept one electron, so two Cl atoms will share an electron pair.
C – will accept four electrons by sharing electrons pairs with hydrogen forming covalent bonds.
H – will donate one electron by sharing an electron pair with carbon.
Question -8
Complete the following:
(a) When the nuclei of two reacting atoms are of _____ mass, then a bond so formed is called _____covalent bond. (Equal, unequal, polar, non -polar).
(b) In case of non-polar covalent bond, the covalent bond is formed in the _____of atoms and shared electrons are distributed _____. (Corner, middle, equally, unequally).
(c) Ionic or electrovalent compounds do not conduct electricity in their …………… state. (Fused/solid)
(d) The ions in ______ compounds are held very strongly due to strong _______ forces. ( electrovalent, covalent, electromagnetic, electrostatic) (2018)
Answer -8
(a) Unequal, polar
(b) Middle, equally
(c) Ionic or electrovalent compounds do not conduct electricity in their solid state.
(d) Electrovalent, electrostatic
Question 9
(a) Compound X consists of molecules. Choose the letter corresponding to the correct answer from the options A, B, C and D given below:
(i) The type of bonding in X will be
A. ionic B. electrovalent C. covalent D molecular
(ii) X is likely to have a
A low melting point and high boiling point
B high melting point and low boiling point
C low melting point and low boiling point
D high melting point and high boiling point
(iii). In the liquid state, X will
A become ionic
B be an electrolyte
C conduct electricity
D not conduct electricity
Answer- 9
(a)
(i) C
(ii) C
(iii) D
Question 10
(a) Electrons are getting added to an element Y:
(i) Is Y getting oxidised or reduced?
(ii) What charge will Y migrate to during the process of electrolysis?
(b) Acids dissolve in water and produce positively charged ions. Draw the structure of these positive ions.
(c) Explain why carbon tetrachloride does not dissolve in water.
Answer 10
(a)
(i) Y is getting reduced.
(ii) Y is positive and it will migrate towards negative electrode that is cathode.
(b) 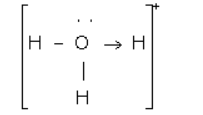
(c) It is anon-polar covalent compound and does not dissolve in polar solvents like water.
Question 11
(a). Elements Q and S react together to form an ionic compound. Under normal conditions, which physical state will the compound QS exist in?
(b). Can Q and S both be metals? Justify your answer.
(c). The property which is characteristic of an electrovalent compound is that
A. it is easily vaporised
B. it has a high melting point
C. it is a weak electrolyte
D. it often exists as a liquid
(d). When a metal atom becomes an ion,
A. it loses electrons and is oxidised
B. it gains electrons and is reduced
C. it gains electrons and is oxidised
D. it loses electrons and is reduced
Answer 11
b. No, in the formation of an ionic compound, one element is a metal and the other is a non-metal.
c. B
d. A
Page-36
Question 12
(a) In the formation of magnesium chloride (by direct combination between magnesium and chloride), name the substance that is oxidized and the substance that is reduced.
(b) What are the terms defined below?
(i) A bond formed by share pair of electrons, each bonding atom contributing one electron to the pair.
(ii) A bond formed by a shared pair of electrons with both electrons coming from the same atom.
Answer 12
(a) Magnesium oxidises and chlorine reduces during the formation of magnesium chloride.
(b) (i) Covalent bond
(ii) Co-ordinate bond
Chapter-2 Chemical Bonding Selina Chemistry Solutions
( Previous Year Solved Question )
Page-36
Question 2009
(a) The one which is composed of all the three kinds of bonds [ionic, covalent and coordinate bonds] is
A. Sodium chloride
B. Ammonia
C. Carbon tetrachloride
D. Ammonium chloride
(b) Draw the structural formula of carbon tetrachloride and state the type of bond present in it.
Answer 2009
(a) Ammonium chloride
(b)
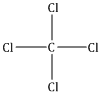
A single covalent bond is present
Question 2010
(a) Select the correct answer fromA, B, C and D. Metals lose electrons during ionisation _____. This change is called
A. Oxidation
B. Reduction
C. Redox
D. Displacement
(b) Select the right answer.
(i) Sodium chloride _______ covalent bond / ionic bond / covalent and coordinate bond.
(ii) Ammonium ion _______ covalent bond / ionic bond / covalent and coordinate bond.
(iii) Carbon tetrachloride _______ covalent bond / ionic bond / covalent and coordinate bond.
Answer 2010
(a) Oxidation
(b)
(i) ionic bond
(ii) covalent and oordinate bond
(iii) covalent bond
Question 2011
(a)
(i) In covalent compounds, the bond is formed due to …………… [sharing/transfer] of electrons.
(ii) Electrovalent compounds have a ……….. [low/high] boiling point.
(iii) . A molecule of ……………. contains a triple bond. [hydrogen, ammonia, nitrogen].
(b) By drawing an electron dot diagram, show the lone pair effect leading to the formation of ammonium ion from ammonia gas and hydrogen ion.
(c) Give reasons. Hydrogen chloride can be termed a polar covalent compound.
Answer 2011
(a)
(i) Sharing
(ii) high
(iii) nitrogen
(b) 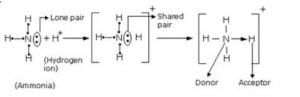
(c) HCl is a covalent compound formed by sharing one electron between chlorine and hydrogen. Because chlorine is more electronegative than hydrogen, the shared pair of electrons shifts towards the chlorine atom. So, a partial negative charge (δ–) develops on chlorine and a partial positive charge (δ+) develops on hydrogen. Hence, the covalent bond is polar in nature.
Question 2012
(a) Draw an electron dot diagram of the structure ofhydronium ion. State the type of bonding present in it.
(b) There are three elements E, F, G with atomic number 19, 8 and 17, respectively. Give the molecular formula of the compound formed between E and G and state the type of chemical bond in this compound.
Answer 2012
(a) 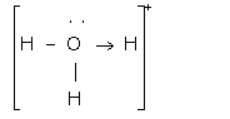
Covalent and coordinate bond
(b) E = 19
F = 8
G= 17
Molecular formula: EG
Chemical bond: Ionic bond
Question 2013
(a) A chemical term for a bond formed by a shared pair of electrons with both electrons coming from the same atom.
(b) Among the compounds, identify the compound that has all three bonds [ionic, covalent and coordinate bond].
A. Ammonia B. Ammonium chloride C. Sodium hydroxide D Calcium chloride
(c) State which is not a typical property of an ionic compound.
A. High m.p.
B. Conducts electricity in molten and aqueous state
C. Are insoluble in water
D. Exist as oppositely charged ions even in the solid state
(d) Compare carbon tetrachloride and sodium chloride with regard to solubility in water and electrical conductivity.
Answer 2013
(a) Dative or coordinate bond
(b) B Ammonium chloride
(c) C Are insoluble in water
(d)
| Carbon tetra chloride |
Sodium chloride |
| It is insoluble in water but dissolves in organic solvents. | this is soluble in water but insoluble in organic solvents. |
| this is a non-conductor of electricity due to the absence of ions. | It does not conduct electricity in the solid state but conducts electricity in the fused or aqueous state. |
Question 2014
(a) Compound ‘X’ consists of only molecules. ‘X’ will have ______
A. Crystalline hard structure B. A lowm.p. and low b.p. C An ionic bond D. A strong force of attraction between its molecules
(b) The molecule which contains a triple covalent bond is
A. ammonia B. methane C. water D. nitrogen
(c) Give one word or phrase for the following: Formation of ions from molecules.
(d) Give reason why covalent compounds exist as gases, liquids or soft solids.
Answer 2014
(a) B
(b) D
(c) Ionisation
(d) Their constituent particles are molecules. These exist as gases or liquids or soft solids because they have weak forces of attraction between their molecules.
Question 2016
(a) The following table shows the electronic configuration of the elements W, X, Y, Z:
| Element | W | X | Y | Z |
| Electronic configurations | 2, 8, 1 | =2, 8, 7 | 2, 5 | 1 |
Answer the following questions based on the table above:
(i) What type of Bond is formed between :
W and X2. Y and Z
(ii) What is the formula of the compound formed between :
X and Z2. W and X
Answer 2016
(i) Ionic bond is formed by transfer of one electron from element W to element X.
(ii) Covalent bond is formed by sharing of electrons between elements Y and Z.
Return to:- Concise Selina Solutions for ICSE Chemistry Class-10
Thanks
Please Share with Your Friends