Water Class-6 Dalal Simplified ICSE Chemistry Solutions Chapter-4, Water Dr Viraf J Dalal Middle School Allied Publishers Solutions. Chapter-4. We Provide Step by Step Solutions of Exercise/Lesson -4 Questions and Answers of Dr Viraf J Dalal Middle School Chemistry Allied Publishers. Visit official Website CISCE for detail information about ICSE Board Class-6.
Water Class-6 Dalal Simplified ICSE Chemistry Solutions Chapter-4
| Board | ICSE |
| Class | 6th |
| Subject | Chemistry |
| Book Name | Dalal New Simplified |
| Chapter-4 | Water |
| Unit-1 | Water |
| Topic | Solution of exercise questions |
| Session | 2023-24 |
Exercise-1
Water Class-6 Dalal Simplified ICSE Chemistry Solutions Chapter-4
Question: 1. State the sources of water –
a] on the earth’s surface
b] below the earth’s surface
Answer: (a) Sources of water on the earth’s surface are —
1. Snow, frost — Snow and frost are the natural forms of water present. They are found in the solid state.
2. Rain water — The purest form of natural water almost free from impurities is rain water. Rain water may dissolve oxygen, nitrogen & carbon dioxide gas forming weak acids. In industrial regions, nitrogen dioxide and sulphur dioxide evolved may dissolve in rain water forming nitric acid & sulphuric acid which causes acid rain.
3. River water — It is one of the impure forms of natural water since most forms of surface water enters into river water. It contains impurities such as sand partides, organic matter, bacteria, mineral salts which dissolve after soil erosion & dissolved gases.
4. Lake water — It is another impure form of natural water which also contains impurities and other soluble salts.
5. Sea water — The most impure form of natural water containing over 3% soluble salts including sodium chloride. It also contains salts of calcium & magnesium.
(b) Sources of Water below the earth’s surface are —
1. Well water — Above the impervious rocky layers, of the earth’s surface is well water. which contains soluble impurities.
2. Spring water — Natural water accumulated above the rocky layers of the earth which forcefully comes out under pressure from the earth’s surface is spring water & contains soluble salts & minerals.
Question: 2. Give the importance of water in –
a] life processes
b] household purpose
c] fire fighting
d] transportation
Answer: The importance of the Water is as follows :
(a) Life processes — Water is used by all plants, animals & humans for carrying out various metabolic processes including photosynthesis by plants & excretion by animals & humans.
(b) Household purposes — Water finds numerous applications, such as watering plants, washing clothes, cooking, bathing, cleaning etc.
(c) Fire fighting — Water is used for extinguishing fires either directly or as a constituent in a fire extinguisher.
(d) Transportation — Water serves as a habitat for marine life i.e. preferred place for an organism to live.
Question: 3. Explain how water plays an important role in –
a] industry
b] agriculture
Answer: (a) Uses of Water in Industry :
1. Water generates electricity in hydroelectric power stations.
2. Water generates steam in boilers, used for various industrial purposes.
3. Water finds application in chemical & other industries for cooling & cleaning operations.
(b) Uses of Water in Agriculture :
In agriculture water finds importance in irrigation, production of crops & as a medium for spraying pesticides.
Question: 4. Give the occurrence of water in the three different states i.e. solid, liquid & gaseous.
Answer: The Occurrence of water in three different states are:
1. Solid state — As snow and frost.
2. Liquid state — In sea water, river water and lake water.
3. Gaseous state — As water vapour in air the amount depending on climatic conditions.
Question: 5. Draw a labelled diagram to show the change of state of water from solid state to liquid state to gaseous state – starting from ice.
Answer: State of water from solid state to liquid state to gaseous state starting from ice:

Question: 6. Explain the term – water cycle. State the main points to show how water moves from the earth’s surface to the atmosphere and back to the earth’s surface as rain.
Answer: The water cycle is a natural process by which the circulation of water takes place from the earth’s surface to the atmosphere and back to the earth’s surface as rain water. The Process of water cycle is discussed below :
FROM THE EARTH’S SURFACE – TO THE ATMOSPHERE
1. Evaporation — The sun’s rays fall on the earth & warm its surface & the air above it. The heat evaporates the water from the streams, rivers & the sea.
2. Water [mainly in the form of water vapour] is also added to the atmosphere by —
(a) Respiration by living organisims Glucose + Oxygen → carbon dioxide + water + energy
(b) Burning of fossil fuels —
Fuel + oxygen → carbon dioxide + water + energy
(3) Formation of clouds — The water vapour along with the warm air rises upwards, where at higher altitudes it condenses into small droplets of water forming clouds.
BACK TO THE EARTH’S SURFACE – AS RAIN WATER
1. Formation of rain — The clouds float in the atmosphere & when the size of the water droplets increases they fall down on the earth as rain water.
2. Rain water falls into streams — The rain water is absorbed by the soil collects underground & flows into streams.
3. Stream water enters rivers & seas — The stream water finds its outlets into rivers & later enters into the seas & oceans.
4. River & sea water evaporates forming clouds and thus continuing the water cycle.
Question: 7. Give a reason why water is considered – a universal solvent.
Answer: Water is universal solvent: Water dissolves many substances forming aqueous solutions. Not only solids, but gases and other liquids also dissolve in water. When water is put in the glass vessel an extremely small amount of glass dissolves in it. Organic compounds [carbohydrates, proteins] also dissolve in water. Hence water is called a universal solvent.
Question: 8. Define the term –
a] solute
b] solvent
c] solution – with reference to addition of sodium chloride to water.
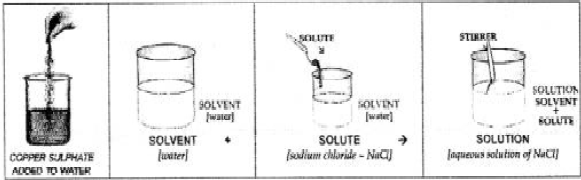
Answer: (a) Solute— The substance which dissolves or disappears in the solvent i.e. liquid to form a solution is called a solute. e.g. sodium chloride.
(b) Solvent— The liquid or medium of dissolution which allows the solute to dissolve in it, so as to form a solution is called a solvent, e.g. water.
(c) Solution— A homogenous mixture of a solute in a solvent is called a solution.
Question: 9. Draw a neat labelled diagram of addition of copper sulphate to water. Label the solute, solvent & solution in the same.
Answer: Diagram of addition of copper sulphate to water. Labt. solute, solvent and solution in the same:
Question: 10. From the following substances given below – state which will form – a homogeneous solution with water.
a] sodium carbonate
b] calcium carbonate
c] charcoal powder
d] sodium sulphate
e] table salt
f] powdered particles of lead
g] iron powder
h] copper filings
i] sand particles
j] honey
Answer: (a) sodium carbonate (d) sodium sulphate (e) table salt (j) honey
Question: 11. Name two gases each –
a] which are soluble in water
b] which are insoluble in water
Answer: (a) Water-soluble gases- Carbon dioxide, ammonia.
(b) Water insoluble gases- Hydrogen, nitrogen.
Question: 12. If ‘X’ g. of potassium nitrate is added to 100 g. of water at 60°C and the salt dissolves completely then –
a] is ‘X’ g. the solubility of potassium nitrate at 60°C.
b] is the solution formed – saturated or unsaturated
c] if on adding ‘X’ + ‘Y’ g. of potassium nitrate to the same amount of water at the same temperature & the solute now just remains behind after stirring then
d] is the solution now – saturated or unsaturated
e] is ‘X’ + ‘Y’ g. the solubility of potassium nitrate.
Answer: Add ‘X’ g. of solute i.e. potassium nitrate to 100 g. of water 60°C.
1. Stir the solute i.e. potassium nitrate in water thoroughly.
2. ‘X’ g. of the solute completely dissolves in water.
3. Add more solute and again stir thoroughly.
4. The solute continues to dissolves.
5. Water i.e. the solvent can dissolve more of the solute at the given temperature.
6. The solution is, therefore, is said to be unsaturated.
Add more solute to water till on adding an amount ‘X + Y’ g. of the solute i.e. potassium nitrate to 100 g. of water at 60°C.
1. The solute just remains behind after stirring.
2. The solution is now saturated.
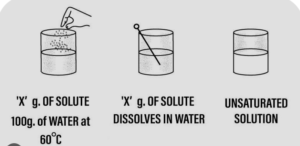
A saturated solution cannot dissolve more of the solute at a given temperature.
Question: 13. State whether the following statements are true or false. If false write the correct statement.
| Statements | True/False |
| a] Solubility of most solids – decrease with increase in temperature. | F |
| b] Distilled water is potable water. | F |
| c] The process to remove germs in water is also called sterilization. | T |
Question: 14. Differentiate between chemical pollution and thermal pollution.
Answer: (a) Chemical pollution: A large number of industrial chemicals which include chemicals from paint, textile & dyestuff industry & various acids & salt solutions enter into the water when discharged as industrial wastes.
Chemical pollutants include — Metallic salt solutions of mercury & lead which cause heavy devastation of marine & plant life.
Agricultural wastes include — Poisonous pesticides namely fungicides & insecticides which may also enter underground water through the soil.
(b) Thermal pollution: Certain industries such as the iron & steel industry & numerous chemical plants use large amounts of water for varied functions.
The discharged wastewater after going through technical processes is rendered hot & on entering streams of natural water enhances the growth of harmful biological organisms.
Question: 15. State some important steps to avoid pollution of water.
Answer: Steps to avoid pollution of water are :
(a) Harmful wastes such as oils and chemicals should not enter the water.
(b) Proper toilets and sewage systems should be used to prevent human excreta, containing disease-causing organisms to enter into the water.
(c) Washing of clothes and bathing should be avoided near water sources.
(d) Planting of trees near water sources including river banks also minimizes pollution.
(e) To minimize thermal pollution the water should be cooled before being discharged as a waste.
(f) Man should be made aware through various awareness programs and media about the harmful effects of water pollution and ways to control it.
Question: 16. State what is meant by the term ‘conservation of water’. State a few water saving methods, which may be used in the home to conserve water.
Answer: Conservation of water is the means of preventing wastage of water so that clean water can be obtained by preventing pollution of water and by protecting the sources of water.
Need for conservation — Inspite of large quantities of water on the earth’s surface only a small percentage is potable water fit for human consumption and household purposes. The need for water is ever increasing and hence all sources of water need to be conserved.
The various methods to conserve the water are :
1. Well should be covered and washing and cleaning should be prevented near a well.
2. Water saving devices must be used in homes
(a) such as closing running taps and using smaller cisterns in toilets.
(b) checking all leakages in household pipes.
(c) turning off the water tap while brushing teeth and while washing hands.
(d) using less electricity, since power plants also consume substantial amount of water.
Question: 17. Give a reason why:
a] Conservation of water is essential in spit of the fact that three fourth of the earth’s surface is covered by water.
b] Polluted water causes disease.
c] Drip irrigation – helps in conservation of water.
Answer: (a) Because only a small percentage is potable water fit for human consumption and household purposes.
(b) Polluted water acts as a carrier for germs which causes various diseases.
(c) Drip irrigation in agriculture utilizes supply of water in small quantities.
—: End of Water Class-6 Dalal Simplified ICSE Chemistry Solutions Chapter-4 :—
Return to – Dalal Simplified Chemistry for ICSE Class-6 Solutions
Thanks
Share with your friends.