Air and Atmosphere MCQs Class-7 Dalal Simplified ICSE Chemistry Solutions Chapter-7 Dr Viraf J Dalal Middle School Allied Publishers Solutions. Chapter-7. We Provide Step by Step Solutions of Exercise/Lesson -7 Match the following, Give reasons, Name of following, Correct answer, Draw diagram of Dr Viraf J Dalal Middle School Chemistry Allied Publishers. Visit official Website CISCE for detail information about ICSE Board Class-7.






Air and Atmosphere MCQs Class-7 Dalal Simplified ICSE Chemistry Solutions Chapter-7
| Board | ICSE |
| Class | 7th |
| Subject | Chemistry |
| Book Name | Dalal New Simplified |
| Chapter-7 | Air and Atmosphere |
| Unit-1 | Air and Atmosphere |
| Topic | Solution of exercise MCQs |
| Session | 2023-24 |
Objective Type Questions
Air and Atmosphere MCQs Class-7 Dalal Simplified ICSE Chemistry Solutions Chapter-7
Question-1. Match the statements in List I with the correct answer from List II.
| List I | List II |
|---|---|
| 1. The constituent of air which is around 0.02% | A: Nitrogen |
| 2. The constituent of air which is inert or unreactive | B: Oxygen |
| 3. The constituent of air which is non-combustible, but supports combustion | C: Sulphur dioxide |
| 4. A pollutant in air responsible for acid rain | D: Carbon dioxide |
| 5. The main rare gas present in air | E: Argon |
Answer-1:
| List I | List II |
|---|---|
| 1. The constituent of air which is around 0.02% | A: Carbon dioxide |
| 2. The constituent of air which is inert or unreactive | B: Nitrogen |
| 3. The constituent of air which is non-combustible, but supports combustion | C: Oxygen |
| 4. A pollutant in air responsible for acid rain | D: Sulphur dioxide |
| 5. The main rare gas present in air | E: Argon |
Question-2. Give reasons for the following:
Question: 1. Acid rain has caused damage to heritage structures such as the Taj Mahal.
Answer: Acid rain damages heritage buildings because it corrodes metallic surfaces and weakens building materials like statues, marbles, limestone. Taj Mahal is predominantly made of lime stone and it reacts with acids present in acid rain.
Question: 2. In the laboratory preparation of oxygen from potassium chlorate – manganese dioxide is added to the reactant.
Answer: Manganese dioxide alters the rate of reaction – KClO3 decomposes at a lower temperature.
Question: 3. Oxygen gas does not affect moist blue litmus paper, but carbon dioxide turns it slightly pink.
Answer: Oxygen is neutral and hence it does not change or react on blue litmus paper, but carbon dioxide is acidic, hence it changes blue litmus slightly pink.
Question: 4. Oxidation of sulphur results in a product which turns moist blue litmus red.
Answer: Oxidation of sulphur results in formation of sulphur dioxide is non-metallic acidic oxide, and hence moist blue litmus turns red.
Question: 5. Combustion and respiration show similarity.
Answer: Combustion and respiration both involve oxidation of carbon and hydrogen to produce carbon dioxide and heat energy.
Question-3. Name the following:
Question: 1. The product formed on oxidation of phosphorus.
Answer: Phosphorus pentoxide
Question: 2. The acid formed when the above product of oxidation of phosphorus, reacts with water.
Answer: Phosphorus acid
Question: 3. The chemical name of rust.
Answer: Hydrated iron (III) oxide (Fe203-xH2O)
Question: 4. The pollutants which combines with water vapor to give sulphuric acid – a product of acid rain.
Answer: Sulphur trioxide
Question: 5. The gas required for both combustion and rusting.
Answer: Oxygen
Question-4. Select the correct answer from the choice in bracket to complete each sentence.
- Nitrogen of the air is utilized by plants for formation of proteins.
- The gas evolved as a product of combustion as well as respiration is carbondioxide.
- A candle burns in ajar of oxygen, but gets extinguished in nitrogen.
- Air has variable composition and hence there is less oxygen at lower altitudes than at higher altitudes.
- Rusting is a chemical reaction which involves slow oxidation.
Question-5. Draw a neat labelled diagram for each of the following experiments.
Question: 1. To determine the component of air used up in burning, by using phosphorus and a bell jar.
Answer:

Question: 2. To study the presence of carbon dioxide.
Answer:

Question: 3. To prove that oxygen is non-combustible, but supports combustion.
Answer:
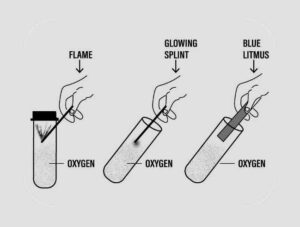
Question: 4. To prepare oxygen in the laboratory from hydrogen peroxide and a catalyst.
Answer:

Question: 5. To demonstrate the burning of sulphur in a jar of oxygen.
Answer:

– : End of Air and Atmosphere Class-7 Dalal Simplified Solutions :–
Return to – Dalal Simplified Chemistry for ICSE Class-7 Solutions
Thanks
Share with your friends.




