Atomic Structure and Chemical Bonding Exe-4B Chemistry Class-9 ICSE Selina Publishers Solutions Chapter-4. Step By Step ICSE Selina Concise Solutions of Chapter-4 Atomic Structure and Chemical Bonding with All Exercise including MCQs, Very Short Answer Type, Short Answer Type, Long Answer Type, Numerical and Structured/Application Questions Solved . Visit official Website CISCE for detail information about ICSE Board Class-9.
Atomic Structure and Chemical Bonding Exe-4B Chemistry Class-9 ICSE Concise Selina Publishers
| Board | ICSE |
| Publications | Selina Publication |
| Subject | Chemistry |
| Class | 9th |
| Chapter-4 | Atomic Structure and Chemical Bonding |
| Book Name | Concise |
| Topics | Solution of Exercise – 4B |
| Academic Session | 2023-2024 |
B. Exercise – 4B
Atomic Structure and Chemical Bonding Class-9 Chemistry Concise Solutions
Page-66
Question 1.
(a) Name the three fundamental particles of an atom.
(b) Give the symbol and charge of each particle.
Answer:
(a) The three fundamental particles of an atom are electron proton and neutron.
(b)
| Particle | Symbol | Charge |
| Electron | e | -1 |
| Proton | p | +1 |
| Neutron | n | No charge |
Question 2.
Complete the table given below by identifying P, Q, R and S.
| Element | Symbol | No. of Protons | No. of neutrons | No. of Electrons |
| Sodium | 11 | P | 11 | |
| Chlorine | Q | 18 | 17 | |
| Uranium | R | 92 | 146 | 92 |
| S | 9 | 10 | 9 |
Answer:
| Element | Symbol | No. of Protons | No. of Neutrons | No. of Electrons |
| Sodium | 11 | 12 | 11 | |
| Chlorine | 17 | 18 | 17 | |
| Uranium | 238U92 | 92 | 146 | 92 |
| S | 9 | 10 | 9 |
Question 3.
The atom of an element is made up of 4 protons, 5 neutrons and 4 electrons. What are its atomic number and mass number?
Answer:
Atomic number = Number of protons or number of electrons = 4
Mass number = Number of protons + Number of neutrons = 4 + 5 = 9
Question 4.
The atomic number and mass number of sodium are 11 and 23 respectively. What information is conveyed by this statement?
Answer:
Atomic number (11) of sodium conveys information that the number of protons and electrons is the same.
Mass number (23) of sodium indicates the sum of protons and neutrons.
Question 5.
Write down the names of the particles represented by the following symbols and explain the meaning of superscript and subscript numbers attached
1p1, 0n1, -1eo
Answer:
p = proton
n = neutron
e = electron
Superscript number : These superscript number show their mass number
Subscript number : These numbers show their atomic number
Question 6.
From the symbol, state the mass number, the atomic number and electronic configuration of magnesium.
Answer:
Mass number = 24
Atomic number = 12
No. of electrons = 24 – 12 = 12
Electronic configuration = 2, 8, 2
Question 7.
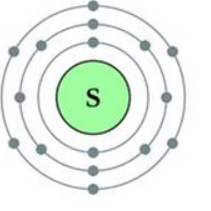
Sulphur has an atomic number 16 and a mass of 32.
State the number of protons and neutrons in the nucleus of sulphur. Give a simple diagram to show the arrangement of electrons in an atom of sulphur.
Answer :
Atomic number = 16
Atomic mass = 32
Number of protons = 16
Number of electrons = 16
Number of neutrons = 32 – 16 = 16
Electronic configuration = 2, 8, 6





Question 8.
Explain the rule according to which electrons are filled in various energy levels.
Answer:
The maximum capacity of a shell to accommodate electrons is given by the general formula 2n2, where n is the serial number of a shell.
The maximum number of electrons possible in the outermost shell is 8 and that in the penultimate shell is 18.
It is not necessary for an orbit to be completed before another is formed. In fact, a new orbit is formed when the outermost shell attains 8 electrons.
Question 9.
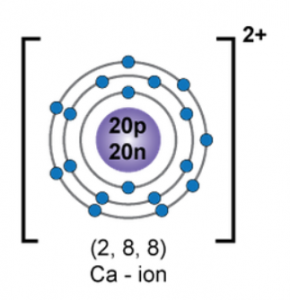
Draw the orbital diagram of ion and state the number of three fundamental particles present in it.
Answer:
Orbital diagram of
Atomic number of Ca2+ is 18.
Electronic configuration of Ca2+ is 2, 8, 8.
 9
9
Number of three fundamental particles of Ca2+:
Protons: 18
Electrons: 18
Neutrons: 40 – 18 = 22
Question 10.
Write down the electronic configuration of the following:
(a) 1327X
(b) 1735Y
Write down the number of electrons in X and neutrons in Y.
Answer:
Number of electrons in X = 13
Mass number of Y = 35 and number of protons in Y = 17
∴ Number of neutrons in Y = 35 – 17 = 18
— : End of Atomic Structure and Chemical Bonding Exe-4B Answer Class-9 ICSE Chemistry Solutions :–
Return to Return to Concise Selina ICSE Chemistry Class-9
Thanks
Please share with your friends




