Goyal Brothers Class-10 Ammonia and Nitric Acid ICSE Chemistry Solutions Ch-9. Step by Step Solutions of Exercise-1 and Exercise -2 , Objective Type Questions of Goyal Brothers Prakashan Chapter-9 Ammonia and Nitric Acid for ICSE Class 10 .
Ammonia and Nitric Acid – test , use, observation seen , preparation , properties (physical and chemical) of Ammonia and Nitric Acid explain with suitable chemical reaction. Visit official Website CISCE for detail information about ICSE Board Class-10 Chemistry .
Goyal Brothers Class-10 Ammonia and Nitric Acid ICSE Chemistry Solutions Ch-9
-: Select Topics :-
Objective Type Questions (update soon)
Exercise 1 Page -161
Goyal Brothers Class-10 Ammonia and Nitric Acid ICSE Chemistry Solutions Ch-9
Question 1. Complete the following chemical equations and balance them.
(i) (NH4)2 SO4 + NaOH ————>
(ii) (NH4)3 PO4 + KOH ———–>
(iii) Mg3 N2 + H2O ———–>
(iv) AIN + H2O ———->
Answer :
(i) (NH)4 SO4 + 2NaOH ————> Na2SO4 + 2NH3 + 2H2O
Ammonium Sulfate + Sodium Hydroxide = Sodium Sulfate + Ammonia + Water
(ii) (NH4)3PO4 + 3KOH → K3PO4 + 3NH3 + 3H2O
Tribasic Ammonium Phosphate + Potassium Hydroxide = Tribasic Potassium Phosphate + Ammonia + Water
(iii) Mg3N2 + 6H2O → 3Mg(OH)2 + 2NH3
Magnesium Nitride + Water = Magnesium Hydroxide + Ammonia
(iv) AIN + 3H2O → NH3 + AI(OH)3
Question 2.
(i) How is ammonia gas prepared in the laboratory, starting from NH4CI?
(ii) Name the substance used for drying ammonia gas.
(iii) Why are substances such as conc. H2SO4, anhydrous calcium chloride and phosphorus pentoxide not used for drying ammonia gas?
Answer :
(i) Preparation in Laboratory
In the laboratory, ammonia is made by heating the ammonium chloride with a quick lime.
2NH4Cl + Ca(OH)2 → CaCl2 + 2H2O + 2NH3
In a rigid glass tube, a mixture of ammonium and quick lime is heated. The received ammonia gas is collected from the gas jar by downward displacement of air.
(ii) drying ammonia gas
the substance used for drying ammonia gas.—Before being collected in a gas jar, ammonia is passed into CaO, which removes the moisture of this gas.
(iii) Concentrated sulphuric acid conc. H2SO4 and anhydrous calcium chloride CaCl2 are not used to dry ammonia because they react with it
Question 3. By stating experimental conditions, briefly describe Haber’s process for the manufacture of ammonia.
Answer :
The Haber Process combines nitrogen from the air with hydrogen to form ammonia. The reaction is reversible and the production of ammonia is exothermic.
N2(g)+H2(g)⇌2NH3(g)
At each pass of the gases through the reactor, only about 15% of the nitrogen and hydrogen converts to ammonia. Gases are cooled and ammonia turns into liquid. Liquid ammonia is separated and rest of the gas is recycled. By continual recycling of the unreacted nitrogen and hydrogen, we get about 98% of ammonia.
Question 4. Why is the heating of catalyst discontinued as soon as nitrogen and hydrogen start reacting during Haber’s process?
Answer :
The electric catalyst in the apparatus is fitted with a heat exchanger
When hydrogen and nitrogen combine ueat is evolved as this reaction is exothermic and therefore the heat exchanger uses this heat to heat the incoming nitrogen and hydrogen gas and external heating is not required
Question 5. Represent Haber’s process by a flowchart.
Answer :
The optimum conditions are temperature=700 K and Pressure=200 atm.
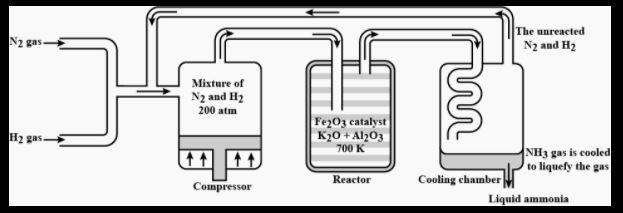





Question 6. Describe an experiment to show the extreme solubility of ammonia gas in water.
Answer :
The fountain experiment allows us to prove the extremely high solubility of ammonia in water. . When the syringe is pushed, water enters the inverted flask containing ammonia gas. This gas is highly soluble thus it dissolves almost immediately in it and reduces the pressure in the flask. As the atmospheric pressure outside is more than the pressure inside the flask, it pushes down on the indicator in the beaker and forces it to enter the flask.
This creates a fountain like feature as it enters the flask and changes colour due to ammonia’s basic nature.
Question 7. Write chemical equations when:
(i) Ammonia gas burns in an atmosphere of oxygen.
(ii) Ammonia gas and oxygen in the form of homogeneous mixture is passed over heated platinum.
(iii) Ammonia gas reacts with excess of chlorine gas.
(iv) Ammonia gas reacts with limited amount of chlorine gas.
Answer :
(i) On burning ammonia in oxygen, the following reaction takes place:
4NH3(g) +3O2(g) 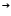 2N2(g) + 6H2O(l)
2N2(g) + 6H2O(l)
(ii)
Catalyst used is Platinum. The catalyst glows since the catalytic oxidation of ammonia is an exothermic reaction. The industrial process is known as Ostwald’s process for the manufacture of nitric acid.
4NH3(g) +5O2(g) 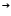 2N2(g) + 6H2O(l)
2N2(g) + 6H2O(l)
(iii)
When the greenish yellow gas chlorine is taken in excess, it reacts with colourless gas ammonia to give a yellow coloured explosive liquid nitrogen trichloride (NCl3).
(iv) Ammonia gas reacts with limited amount of chlorine gas
Produced hydrogen chloride vapor can behave as an acidic compound (can release H+ ions in the water). Then, hydrogen chloride reacts with basic ammonia gas to produce ammonium chloride which is a solid white smog. To happen this second step reaction, ammonia is require
Question 8. Describe an experiment to show the reducing nature of ammonia gas.
Answer :
Ammonia reduces CuO to Cu.
Question 9. Describe all what you will observe and write chemical equations, when a limited amount of ammonia gas is passed through the following aqueous solutions:
(i) ZnCl2 (ii) FeSO4 (iii) FeCl3 (iv) Pb (NO3)2 (v) Cuso4.
Answer :
when limited amount of ammonia gas is passed through the following aqueous solutions:
(i) ZnCl2
Ammonia gas on reacting with aqueous solution of zinc chloride produces white gelatine like precipitate of zinc hydroxide.
ZnCl2 + 2 NH4OH → Zn (OH)2
(ii) FeSO4
Ammonia gas on reacting with aqueous solution of ferrous sulphate produces dirty green precipitate of ferrous hydroxide.
FeSO4 + 2 NH4OH →(NH4)2SO4 + Fe(OH)2
(iii) FeCl3
(iv) Pb (NO3)2
When ammonia is passed through an aqueous solution of lead nitrate, chalky white precipitate of lead hydroxide is formed which is insoluble in excess of ammonia.
Pb(NO3)2+2NH4OH→2(NH4)NO3+Pb(OH)2↓
(v) Cuso4.
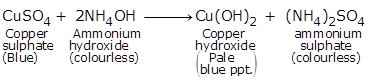
Question 10. State four industrial uses of ammonia gas,
Answer :
Ammonia is also used as a refrigerant gas, for purification of water supplies, and in the manufacture of plastics, explosives, textiles, pesticides, dyes and other chemicals. It is found in many household and industrial-strength cleaning solutions
Question 11. State two tests for ammonia gas.
Answer :
When added to a solution of copper sulphate, ammonia turns the solution deep blue.
NH3 + CuSO4 + nH2O→ [Cu(NH3)4(H2O)n]SO4
When added to Nessler’s reagent (basic arrangement of K2[HgI4], ammonia gives a precipitate brown in colour.
NH4+ + 2[HgI4]2− + 4OH− → HgO·Hg(NH2)I ↓ + 7I− + 3H2O
Question 12. What properties of ammonia gas make it suitable as a refrigerant in cold storages?
Answer :
It has a low boiling point and is favoured since i) highly energy-efficient refrigerant, having minimal impact on environment ii) having zero Ozone Depletion Potential (ODP) iii) having zero Global Warming Potential (GWP)
Question 13. What is liquor ammonia? How does it differ from liquid ammonia?
Answer :
Both liquid and liquor ammonia are in the liquid state. However, the liquid ammonia has only ammonia molecules, but the liquor ammonia has both ammonia and water. The key difference between liquid ammonia and liquor ammonia is that the liquid ammonia contains NH3 molecules, while the liquor ammonia contains NH4OH
Exercise 2 Page – 170 to 171
Goyal Brothers Class-10 Ammonia and Nitric Acid ICSE Solutions Ch-9
Question 1.
(a) How is nitric acid prepared in the laboratory?
(b) Why should the apparatus used to prepare nitric acid be of all glass?
(c) Why is the nitric acid collected above pale yellow in colour?
(d) Why should the temperature of the reaction mixture of nitric acid not be allowed to rise above 200 ° C?
Answer :
(a) In laboratory, nitric acid can be made by thermal decomposition of copper(II) nitrate, producing nitrogen dioxide and oxygen gases, which are then passed through water to give nitric acid.
(b) The apparatus used for the laboratory preparation of nitric acid are made of glass completely because the vapours of nitric acid are highly corrosive. They attack rubber cork, plastics etc.
(c) Nitric acid obtained in laboratory is slightly yellow due to the dissolution of nitrogen dioxide which is produced due to thermal decomposition of a portion of HNO3.
(d) At high temperature, nitric acid decomposes and the glass apparatus may get damaged. Sodium formed at a higher temperature forms a hard crust which sticks to the walls of the retort.
Question 2.
(a) How is nitric acid prepared in large scale?
(b) What is the use of catalytic chamber in the above process?
(c)Why is nitric oxide passed through a heat exchanger before transferring to another oxidising tower in the above process
(d) What is the function of absorption tower in the above process?
Answer :
(a) In industries nitric acid is prepared by Ostwald process.
NH3 + O2 ——> NO + H2O
Catalyst is Platinum at 800C temperature.
NO + O2 ——> NO2
NO2 + H2O + O2 ——-> HNO3
(b) This chamber uses a catalyst-like platinum gauze or copper and nickel can also be used. The oxidation of ammonia takes place and it is converted into nitric oxide (NO). The process is reversible and exothermic. The change in temperature encourages a forward reaction
(c)
The nitric oxide gas produced by oxidation of ammonia is in a very hot state. It is passed through a heat exchanger in which the temperature of nitric oxide is lowered to 150oC. After cooling, nitric oxide is transferred to another oxidizing tower where nitrogen dioxide (NO2) is oxidized at about 50oC.
2NO + O2 ↔2 NO2
(d) In a special absorption tower containing water, the nitrogen dioxide from the secondary oxidation chamber is introduced. NO2 gas is passed through a tower where it absorbs the water. Nitric acid is then obtained through this process. Nitric acid that is obtained is very dilute
Question 3. Write fully balanced equations for the reaction of dilute nitric acid with the following chemicals:
(i) Sodium bicarbonate (ii) Calcium hydroxide (iii) Lead monoxide
(iv) Zinc carbonate (v) Sodium hydroxide (vi) Copper metal
Answer
reaction of dilute nitric acid with the following chemicals:
(i) Sodium bicarbonate
Na2CO3 + 2HNO3 = 2NaNO3 + CO2 + H2O.
(ii) Calcium hydroxide
2HNO3(aq.)+ CaO(s) —>Ca(NO3)2(aq.)+ H2O(l).
(iii) Lead monoxide
PbO + 2 HNO3(concentrated) → Pb(NO3)2↓ + H2O.
(iv) Zinc carbonate
ZnCO3 + 2HNO3 → Zn(NO3)2 + CO2 + H2O.
(v) Sodium hydroxide
HNO3 + NaOH = NaNO3 + H2O
(vi) Copper metal
4 HNO3(l) + Cu(s) ==> Cu(NO3)2(s and aq) + 2 NO2(g) + 2 H2O(l)
Question 4. Write fully balanced equations for the reaction of dilute nitric acid with the following chemicals:
(i) Charcoal
(ii) Sulphur
(iii) Phosphorus
(iv) Copper
Answer :
balanced equations for the reaction of dilute nitric acid
(i) Charcoal
(ii) Sulphur
S+6HNO3(hot & conc)→H2SO4+2H2O+6NO2
(iii) Phosphorus
P + 5HNO3. H3PO4 + 5NO2 + H2O
(iv) Copper
4 HNO3(l) + Cu(s) ==> Cu(NO3)2(s and aq) + 2 NO2(g) + 2 H2O(l)
Question 5. State four industrial uses of nitric acid.
Answer :
- Industrial Uses of Nitric Acid-It is used in manufacturing several types of polymers like polyamides and polyurethane. Nitric acid is also commonly used as rocket propellants in the aerospace industry
- fertilizer production, Nitric acid is used for manufacturing different types of nitrogenous fertilizers like calcium nitrate, ammonium nitrate, etc. Nitric acid is key component which is also a by product of Ammonia
- used for manufacturing nitrogen-based compounds like nylon as well as most of the explosives like trinitrotoluene (T.N.T.), nitroglycerin, amongst others
- It is also used in food industries and helps in cleaning food and equipment, etc.
Question 6. State two tests for dilute nitric acid.
Answer :
(1). On reacting copper metal with nitric acid brown fumes of nitrogen dioxide.
4 HNO3(l) + Cu(s) ==> Cu(NO3)2(s and aq) + 2 NO2(g) + 2 H2O(l)
(2) Brown ring test : Nitric acid in a test tube is taken and then and then freshly prepared ferrous sulphate solution is added. Concentrated H2SO4 is then added carefully down the sides of the test tube, dark brown ring is formed at the juctions of two layers
2HNO3+ 3H2SO4 + 6FeSO4 —>> 3Fe2(SO4)3 + 2NO + 4H2O
Question 7. What will you observe, when the following nitrates are heated strongly. Support your answer with fully balanced chemical
(i) Ammonium nitrate (ii) Sodium nitrate (iii) Zinc nitrate (iv) Lead nitrate (v) Copper nitrate (vi) Mercuric nitrate.
equations
Answer :
Observation , when the following nitrates are heated strongly.
(i) Ammonium nitrate
Ammonium Nitrate (NH4NO3) when heated gives Nitrous Oxide (N2O) also called laughing gas and Water (H2O)
(ii) Sodium nitrate
Sodium nitrate (and potassium nitrate) upon heating decomposes to produce sodium nitrite and colorless and odourless oxygen gas. This characterizes the colorless oxygen gas as it rekindles a burning wooden splinter. Sodium nitrite (and potassium, naturally) is resistant to heat
(iii) Zinc nitrate
When zinc nitrate crystals are strongly heated, zinc oxide white residue and brown vapors of nitrogen dioxide gas are formed
(iv) Lead nitrate
When lead nitrate is heated in a test tube, a brownish yellow gas is formed which is heavier than air. The gas is nitrogen dioxide. The products of the reaction are lead oxide, nitrogen dioxide and oxygen
(v) Copper nitrate
When copper nitrate heated first heat losses the water molecule, on further heating it decomposes to give reddish brown gas Nitrogen dioxide and oxygen. The greenish blue crystals of copper nitrate will change to black residue of copper oxide and give reddish brown gas i.e., nitrogen dioxide on heating
(vi) Mercuric nitrate.
When lead nitrate is heated in a test tube, a brownish yellow gas is formed which is heavier than air. The gas is nitrogen dioxide. The products of the reaction are lead oxide, nitrogen dioxide and oxygen
Question 8. Describe two tests for nitrates.
Answer :
(1). On reacting copper metal with nitric acid brown fumes of nitrogen dioxide.
4 HNO3(l) + Cu(s) ==> Cu(NO3)2(s and aq) + 2 NO2(g) + 2 H2O(l)
(2) Brown ring test : Nitric acid in a test tube is taken and then and then freshly prepared ferrous sulphate solution is added. Concentrated H2SO4 is then added carefully down the sides of the test tube, dark brown ring is formed at the juctions of two layers
2HNO3+ 3H2SO4 + 6FeSO4 —>> 3Fe2(SO4)3 + 2NO + 4H2O
Objective Type Questions Page 171 to 174
I. Multiple Choice Questions
Goyal Brothers Class-10 Ammonia and Nitric Acid ICSE Solutions Ch-9
Choose the correct answer from the options given below :
update soon
- …………………
- ………………….
- ………………….
- ……………………
- ……………………
- ……………………
- ……………………
- ……………………
- ……………………
- ……………………
- ……………………
- ……………………
- ……………………
- ……………………
- ……………………
- ……………………
- ……………………
- ……………………
- ……………………
- ……………………
- ……………………
- ……………………
- ……………………
- ……………………
- ……………………
- ……………………
- ……………………
- ……………………
- ……………………
- ……………………
- ……………………
- ……………………
- ……………………
- ……………………
- ……………………
- ……………………
- ……………………
- ……………………
II. Fill in the blanks spaces with the choice given in brackets:
- …………………..
- ……………………..
- …………………
- …………………
- ………………..
- ………………..
- …………………………..
- …………………………
- ………………………….
- ……………………………..
- …………………………….
- ……………………
- ……………………
- ……………………
- ……………………
- ……………………
- ……………………
- ……………………
- ……………………
- ……………………
III. Choose from the following list, as what matches the descriptions given below.
[Copper oxides, Iron (III) hydroxide, Haber’s process, ammonia, potassium mercury iodide, calcium hydroxide, ammonium nitrate, KNO3, KNO2, Pb(NO3)2 NO2, Magnesium, Hg (NO3)2, bluish green, Zn (NO3)2]
1. A base in solid form used in the laboratory preparation of ammonia gas.
2. A black colour oxide of metal changes to reddish colour when heated and ammonia gas passed over it.
3. The name of chemical dissolved in Nesslar’s reagent.
4. A reddish brown precipitate formed when ammonia gas is passed through Iron (III) sulphate solution.
5. An ammonium salt which decomposes explosively to form nitrous oxide gas and water on heating.
6. A process used in the synthesis of ammonia gas.
7. A gas used in the manufacture of nitric acid by Ostwald’s process.
8. A nitrate commonly used in the laboratory preparation of nitric acid.
9. A gas which stains nitric acid yellow during its laboratory preparation.
10. A metal which reacts with very dilute nitric acid to liberate hydrogen gas.
11. A nitrate of a metal which decomposes to form a nitrite and oxygen gas.
12. A nitrate which decomposes with decripitating sound and leaves behind a residue which is reddish brown when hot and yellow when cold.
13. A nitrate which decomposes on heating to form a residue white yellow when hot and white when cold.
14. A nitrate of metal which on heating leaves behind pure metal as residue.
15. The colour of reaction mixture when conc. nitric acid is poured over copper turnings.
–: end of Ammonia and Nitric Acid Solutions Goyal Brothers :–
Return of : Chemistry Class-10 Goyal Brothers Prakashan
Thanks
Share with your friends

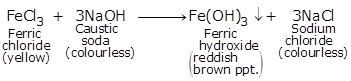




4 thoughts on “Goyal Brothers Class-10 Ammonia and Nitric Acid ICSE Chemistry Ch-9”
For all chapter you are not uploading mcq ans kindly upload it
Do yourself if feel difficulty call 8957797189
Where is mcq ans
Do yourself if feel difficulty call 8957797189