Metallurgy Dalal Simplified ICSE Chemistry Class-10 Solutions Chapter 6 . Solutions of Dr Dalal Simplified ICSE Chemistry by Dr Viraf and J Dalal for Class 10. Step by step Solutions of Metallurgy ICSE Chemistry by Dr Viraf and J Dalal Simplified Chemistry.
Metallurgy Dalal Simplified ICSE Chemistry Class-10
Get Other Chapter Dalal Simplified ICSE Chemistry Class-10 Solutions
How to Solve Mole Concept ICSE Chemistry Class-10
Note:– Before viewing Solutions of Metallurgy by Dr Viraf and J Dalal Simplified ICSE Chemistry Solutions of Chapter-6 .Read the Chapter-6 Metallurgy Carefully to understand the concept in better way .After reading the Chapter-6 Metallurgy solve all example of your text book with ICSE Specimen Sample Paper for Class-10 Exam of Council. Focus on Metallurgy Concept is the Most important Chapter in ICSE Class 10 Chemistry.Previous Year Solved Question Paper for ICSE Board
Additional Questions Metallurgy Dalal Simplified ICSE
Question 1.
State how the physical and chemical property differences between metals and non-metals are related to their basic atomic structure.
Define a metal with particular emphasis on (i) ionization (ii) valency (iii) formation of oxides.
State the position [group] in the periodic table to which the following metals belong (i) Na – alkali metal (ii) Mg – alkaline earth metal (iii) Fe and Zn transition elements (iv) inner transition elements (metals) (v) Al – post transition element.
Answer:
Atoms of Metallic elements, in general, have relatively larger atomic size. Hence, their valence electrons are held less tightly by the nucleus. As such, the valence electrons are mobile, leading to good electrical and thermal conductivity in case of metals. Further, with 1,2 or 3 valence electrons, metals can easily lose electrons to form cations. Thus, metal are electropositive in nature and are strong reducing agents.
On the other hand, non-metallic elements, in general, have relatively smaller atomic size. Hence their valence electrons are held tightly by the nucleus. As such their valence electrons are not mobile, thus making non-metals bad conductor of heat and electricity. Further, non-metals have 5, 6 or 7 valence electrons. As such, non-metals tend to gain electrons to form anions. Thus non-metals are electronegative in nature and strong oxidising agents.
Thus, we find that the physical and chemical properties of metals and non-metals are related to their basic atomic structure.
Defination of Metals :
- Atoms of metals with 1, 2 or 3 electrons in their valence shell can easily lose their valence electrons to form cations e.g.
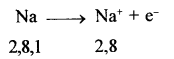





- As metals can easily lose electrons to form cations, metals are electropositive in nature.
- Metals on reacting with non-metals form ionic or electrovalent compounds, e.g.

- As metals tend to form cations, metals show a positive valency of 1,2 or 3.
Position of Metals in the periodic table :
- Na – alkali metal – Group I (IA)
- Mg – alkaline earth metal – Group 2 (IIA)
- Fe and Zn transition elements – Group 9 (VIII), Group 12 (IIB)
- Inner transition elements : zero group.
- Al-post transition element: Group 13 (III A)
Question 2.
Metals occur in the free state and in the combined state, name two metals which occur in the free or native state. In the combined state metals occur in the form of compounds. Name two different metallic compounds in each case which occur as
- halides
- oxides
- sulphides.
Answer:
Gold and Platinum are the two metals that occur in the free or native state.
The metallic compounds which occur in :
- Halides – Cryolite[Na3AlF6], Flurospar[CaF2], Rock Salt[NaCl]
- Oxides – Bauxite[Al2O3.2H2O], Zincite[ZnO], Cuprite[Cu2O]
- Sulphides – Iron Pyrite[FeS2], Zinc blende[ZnS], Gdlena[PbS]
Question 3.
Differentiate between
- mineral & ore
- matrix & flux.
Answer:
Differentiate between mineral & ore
Mineral :
- The compounds of various metal found in nature associated with their earthly impurities are called minerals.
Ore :
- The naturally occurring minerals from which metals can be extracted profitably and conveniently are called ores,
Differentiate between matrix & flux
- Matrix : The rocky impurities including silica [SiO2], mud etc. associated with the ore is called matrix or gangue.
- Flux : The substance added to the ore to get rid of the matrix resulting in the formation of a fusible compound slag.
Question 4.
Give the (i) common (ii) chemical nadie (iii) formula of two common ores each of aluminium, zinc and iron.
Answer:
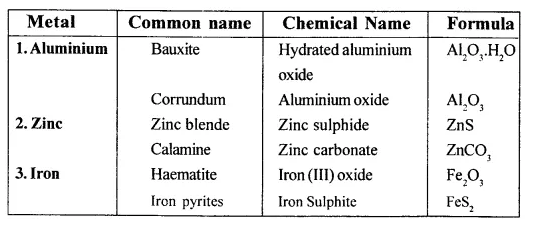
Question 5.
In the stages involved in the extraction of metals in general – give reasons for the following.
(i) Dressing of the ore is an essential process in the extraction of metal from its ore.
Answer:
The ores are found mixed with earthy impurities like sand, clay, lime stone etc. These unwanted impurities in the ore are called gangue or matrix.
The process of removal of gangue from powdered ore is called concentration or ore dressing.
Hence, it is the essential process in the extraction of metal from its ore because it convert’s the impure ore to pure concentrated ore
(ii) An electromagnetic wheel is used in the magnetic separation process of ore from gangue.
Answer:
An electromagnetic wheel is used in the magnetic separation process of ore from gangue as it seperate’s the magnetic particles from the non-magnetic particles. The magnetic particles get attracted to the magnetic wheel and thus get seperated from the gangue.
(iii) In the froth floatation process, the ore floats on the top & the gangue settles down.
Answer:
The impurities get wetted by water and remain behind in the tank. Since, the ore is lighter, it comes on the surface with the froth and the impurities(gatlgue) are left behind.
(iv) Magnetic separation is not used during the dressing of bauxite ore in the extraction of aluminium.
Answer:
Bauxite is concentrated by Leaching (Baeyer’s process). The impure bauxite is treated with concentrated NaOH, Al2O3 and SiO2 dissolve, but Fe2O3 and other basic materials remain insoluble and are removed by filtration.
Aluminium is highly reactive metal, belonging to the III A group of the periodic table. In nature, aluminium is found in the form of its oxide in its ore.
Hence, Magnetic separation is not used during the dressing of bauxite ore in the extraction of aluminium.
(v) Conversion of concentrated ore to its oxide is an essential step in the extraction of metals from the ore, even then the step is not necessary in the metallurgy of aluminium.
Answer:
Conversion of concentrated ore to its oxide is an essential step in the extraction of metals from the ore, even then the step is not necessary in the metallurgy of aluminium because in metallurgy of aluminium, the ore is already an oxide.
(vi) Roasting of the concentrated ore is carried out in the presence of excess air, while calcination of the concentrated ore in the absence or limited supply of air.
![]()
Answer:
Roasting is a process of converting an ore into its oxide by heating strongly in presence of excess air, so that oxygen gets added to form the corresponding oxide. It is done on sulphide ores in order to remove sulphur as sulphur gets escape in the form of gas.
Whereas in carbonates ores, one needs to drive out carbonate and moisture impurities. So, ore is heated to a high temperature in absence of air. This process is called calcination.
![]()
(vii) Roasting the ore generally results in evolution of sulphur dioxide gas, while calcination of the ore evolves carbon dioxide gas.
Answer:
Generally, sulphide ores are roasted, so SO2 is given off.
While carbonate and hydrated ores are calcined, so CO2 or water vapours are given off.
(viii) Reduction of metallic oxides to metal in the extraction of metals from the ores- is based on the position of the metal in the activity series.
Answer:
The method used to extract a metal from its ore depends on the position of the metal in the reactivity series.
(a) Metals higher up in the series need to be extracted using electricity.
(b) Metals lower in the series can be extracted by reduction with carbon.
(ix) Oxides or halides of highly electropositive metals e.g. K, Na, Ca, Al are reduced to metals by electrolysis and not by reduction with coke.
Answer:
Oxides are highly active metals like potassium, sodium, calcium, magnesium and aluminium have great affinity towards oxygen and so cannot be reduced by coke.
(x) Coke is not preferred as a reducing agent in the reduction of the oxide of mercury to its metal, but is preferred in the reduction of the oxide of zinc to its metal.
Answer:
Coke is not preferred as a reducing agent in the reduction of the oxide of mercury to its metal because metals low in the activity series are very reactive and the oxides of the metals can be reduced to metals by heating in air.
The reducing nature of carbon plays an important role in metallurgy and helps extract metals from their ores.
For example :
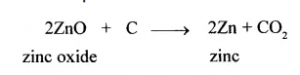
Question 6.
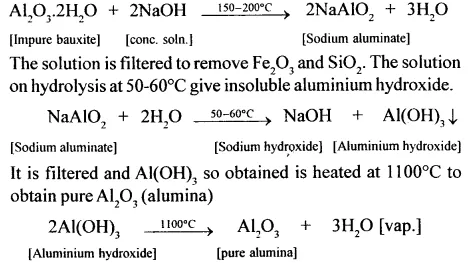
In the extraction of aluminium from bauxite, the first step is the dressing of the ore by Baeyer’s process. Give balanced equations for the conversion of impure bauxite to pure alumina using a concentrated solution of NaOH.
Answer:
Bauxite (red) contains Fe2O3. and SiO2 as the main impurities. It is crushed to a powder and is treated with a cone, solution of NaOH at 150-200°C (under pressure) when Al2O3 dissolves.
Question 7.
In the electrolytic reduction of pure alumina to pure aluminium – by Hall Herault’s process, give the electrolytic reactions involved in the same, resulting in formation of aluminium at the cathode.
Answer:
Aluminium is obtained from alumina (Al2O3) by electrolytic reduction. The electrolytic both consists of a mixture of cryolite (Na3AlF6) and fluorspar (CaF2) and alumina (Al2O3)

Question 8.
State the function of
- NaOH
- cryolite
- fluorspar in the metallurgy of aluminium.
Answer:
Functions of :
(i) NaOH : Crushed and powdered bauxite is heated with a cone, solution of NaOH at 150-200°C for 2 hours under pressure. The main impurities present in bauxite (Fe2O3 and SiO2) remain unaffected with cone. NaOH since these are not amphoteric. Bauxite, being amphoteric reacts with the base to form sodium aluminate, which is soluble in water. Thus NaOH helps in the purification of the ore.
(ii) Cryolite :
(a) To increase the mobility of the fused mixture.
(b) To lower the fusion point of the mixture.
(c) To increase the electrical conductivity of the mixture.
(iii) Fluorspar :
(a) To increase the mobility of the fused mixture.
(b) To lower the fusion point of the mixture.
Question 9.
Give reasons for the following – pertaining to Hall Herault’s process.
- The fusion temperature of the electrolyte has to be lowered before conducting the electrolytic reduction.
- The constituents of the electrolyte in addition to one part of fused alumina contains three parts of cryolite and one part of fluorspar.
- A layer of powdered coke sprinkled over the electrolytic mixture, protects the carbon electrodes.
- It is preferred to use a number of graphite electrodes as anode, instead of a single graphite electrode.
Answer:
(i) The liberated A1 metal [m.p. 660°C] may also tend to volatize out and get wasted. Hence the fusion temperature of the electrolytic mixture has to be – lowered.
(ii) The reasons for addition of mainly cryolite [in a higher ratio] and fluorspar to the electrolytic mixture are :
(a) They lowers the fusion point of the mixture i. e. the mixture fuses [melts] around 950°C instead of 2050°C.
(b) They enhances the mobility of the fused mixture by acting as a solvent for the electrolytic mixture.
Thus cryolite in the molten state of subdivision dissolves aluminium oxide.
(c) Addition of cryolite enhances the conductivity of the mixture.
Since pure alumina is almost a non-conductor of electricity
(iii) The layer of powdered coke is sprinkled over the electrolytic mixture because :
(a) It prevents burning of carbon electrodes in air at the emergence point from the bath.
(b) It minimizes or prevents heat loss by radiation.
(iv) The graphite[carbon] anodes are continously replaced during the electrolysis because :
(a) The oxygen evolved at the anode escapes as a gas or reacts with the carbon anode.
(b) The carbon anode is thus oxidised to carbon monoxide which either bums giving carbon dioxide or escapes out through an outlet.
2C + O2 → 2CO [2CO + O2 → 2CO2]
(c) The carbon anode is hence consumed and renewed periodically after a certain period of usage,
Question 10.
Define — (a) alloy, (b) amalgam
Answer:
- Alloy : An alloy is a homogenous mixture of two or more metals and non-metals, out of which at least one is a metal.
For example, brass (Zn + Cu), bronze (Cu + Sn), solder (Sn +Pb), stainless steel (Fe + Cr + Ni + C) etc. - Amalgam : An alloy in which mercury is an essential constituent is called an amalgam.
For example, Dental amalgam (Hg + Ag + Sn), it is used for filling dental cavities.
Question 11.
State (i) composition (ii) reason for alloying (iii) one use of each of the following alloys.
- Brass
- Bronze
- Duralumin
- Solder [fuse metal]
- Stainless steel
Answer:

–Try Also :–




