Nootan Solutions Heat Engine Second Law of Thermodynamics ISC Physics Class-11 Ch-21 Vol-2 Nageen Prakashan. Step by step Solutions of Numericals of latest edition of Kumar and Mittal ISC Physics Part-2 Class-11 Nageen Prakashan Questions. Visit official Website CISCE for detail information about ISC Board Class-11 Physics.
Nootan Solutions Heat Engine Second Law of Thermodynamics ISC Physics Class-11 Ch-21 Vol-2 Nageen Prakashan.
| Class: | 11 |
| Subject: | Physics Part-2 |
| Chapter | 21 Heat Engine Second Law of Thermodynamics |
| Board | ISC |
| Writer / Publications | Nootan / Nageen Prakashan / Kumar and Mittal |
| Topics | Solved Numericals of page 798,799 |
Nootan Solutions Heat Engine Second Law of Thermodynamics ISC Physics Class-11 Ch-21 Vol-2 Kumar and Mittal
The second law of thermodynamics gives a fundamental limitation to the efficiency of a heat engine and the coefficient of performance of a refrigerator. It says that efficiency of a heat engine can never be unity (or 100%). This implies that heat released to the cold reservoir can never be made zero.
This principle which disallows certain phenomena consistent with the First law of thermodynamics is known as the second law of thermodynamics.
Following are the two statements of second law of thermodynamics.
Kelvin-Planck Statement: It is impossible to construct an engine, operating in a cycle, to extract
heat from hot body and convert it completely into work without leaving any change anywhere i.e., 100% conversion of heat into work is impossible.
Clausius Statement: It is impossible for a self acting machine, operating in a cycle, unaided by any external energy to transfer heat from a cold body to a hot body. In other words heat can not flow itself from a colder body to a hotter body.
heat engine
A heat engine is a device by which a system is made to undergo a cyclic process that results
in conversion of heat to work. Basically, a heat engine consists of:
(i) a heat source,
(ii) a heat sink, and
(iii) a working substance.
Carnot’s Engine.
He proposed a hypothetical engine working on a cyclic/reversible process operating between two temperatures. Its efficiency is independent of the working substance and is given by, η=1-T2/T1 where T1 is the temperature of source and T2 is the temperature of sink.
According to Carnot’s theorem:
(a) working between two given temperatures T1 and T2 of the hot and cold reservoirs respectively, no engine can have efficiency more than that of Carnot’s engine, and
(b) the efficiency of the Carnot engine is independent of the nature of the working substance.
Refrigerator
The process of removing heat from bodies colder than their surroundings is called refrigeration and the device doing so is called refrigerator.
In the refrigerator, heat is absorbed at low temperature and rejected at higher temperature with the help of external mechanical work. Thus, a refrigerator is a heat engine working backward and hence it is also called heat pump.
Refrigerator works on the reverse process of Carnot engine. By the work done on the system, heat is extracted from low temperature sink T2 and passed on to high temperature source T1. The coefficient of performance is given by
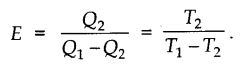





Nootan Solutions Heat Engine Second Law of Thermodynamics ISC Physics Class-11 Ch-21 Vol-2
(Page-798)
CONTACT FOR LIVE CLASSES- 9335725646
-: End of Heat Engine Second Law of Thermodynamics Nootan Solutions :-
Return to – Nootan Solutions for ISC Physics Class-11 Nageen Prakashan
Thanks
Share with your friends




