Study of Gas Laws Exe-7 Numericals Answer Chemistry Class-9 ICSE Selina Publishers Solutions Chapter-7. Step By Step ICSE Selina Concise Solutions of Chapter-7 Study of Gas Laws with All Exercise including MCQs, Very Short Answer Type, Short Answer Type, Long Answer Type, Numerical and Structured/Application Questions Solved . Visit official Website CISCE for detail information about ICSE Board Class-9.
Study of Gas Laws Exe-7 Numericals Answer Chemistry Class-9 ICSE Concise Selina Publishers
| Board | ICSE |
| Publications | Selina Publication |
| Subject | Chemistry |
| Class | 9th |
| Chapter-7 | Study of Gas Laws |
| Book Name | Concise |
| Topics | Solution of Exercise – 7 NumericalsAnswer Type |
| Academic Session | 2023-2024 |
F. Exercise – 7 Numericals Answer Type
Study of Gas Laws Class-9 Chemistry Concise Solutions
Page-133
Question 1.
Convert :
(i) 273°C to kelvin
(ii) 293 K to °C
Answer:
(i) Kelvin = °C + 273 = 273 + 273 = 546 K
(ii) °C = Kelvin – 273 = 293 – 273 = 20°C
Question 2.
What will be the minimum pressure required to compress 500 dm3 of air at 1 bar to 200 dm3 temperature remaining constant.
Answer:
V1 = 500 dm3
P1 = 1 bar
T1 = 273 K
V2 = 500 dm3
T2 = 273 K
P2= ?






Question 3.
2 litres of a gas is enclosed in a vessel at a pressure of 760 mmHg. If temperature remains constant, calculate pressure when volume changes to 4 dm3.
Answer:
V = 2 litres
P = 760 mm
V1 = 4000 m3 [1 dm3 = 4 litres]
P1= ?
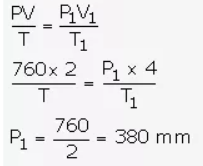
Question 4.
800 cm3 of gas is collected at 654 mm pressure. At what pressure would the volume of the gas reduce by 40% of its original volume, temperature remaining constant?
Answer:
Given:
V = 800 cm3
P = 650 m
P1= ?
V1 = reduced volume = 40% of 800
= 8000× 40/100= 320
Net V1 = 800 – 320 = 480 cm3
T = T1
Using the gas equation,

Since T = T1

=1083.33 mm of Hg.
Question 5.
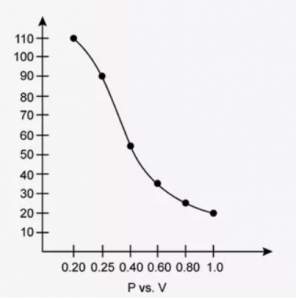
At constant temperature, the effect of change of pressure on volume of a gas was as given below:
| Pressure in atmosphere | Volume in litres |
| 0.20 | 112 |
| 0.25 | 89.2 |
| 0.40 | 56.25 |
| 0.60 | 37.40 |
| 0.80 | 28.10 |
| 1.00 | 22.4 |
(a) Plot the following graphs
- P vs V
- P vs 1/V
- PV vs P
Interpret each graph in terms of a law.
(b) Assuming that the pressure values given above are correct, find the correct measurement of the volume.
Answer:
At constant temperature, the effect of change of pressure on volume of a gas was as given below:
| Pressure in atmosphere | Volume in litres |
| 0.20 | 112 |
| 0.25 | 89.2 |
| 0.40 | 56.25 |
| 0.60 | 37.40 |
| 0.80 | 28.10 |
| 1.00 | 22.4 |
(a) Plot the following graphs
- P vs V
2. P vs 1/V
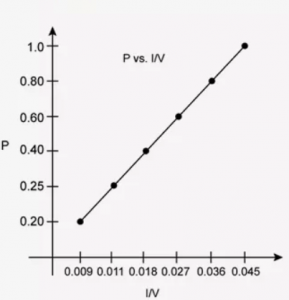
3. PV vs P

Interpret each graph in terms of a law.
(b) Assuming that the pressure values given above are correct, find the correct measurement of the volume.

Question 6.
A cylinder of 20 litres capacity contains a gas at 100 atmospheric pressure. How many flasks of 200 cm3capacity can be filled from it at 1 atmosphere pressure, temperature remaining constant?
Answer:

Question 7.
A steel cylinder of internal volume 20 litres is filled with hydrogen at 29 atmospheric pressure. If hydrogen is used to fill a balloon at 1.25 atmospheric pressure at the same temperature, what volume will the gas occupy?
Answer:
V = 20 litre
P = 29 atm
P1 = 1.25 atm
V1 =?
T = T1

Question 8.
561 dm3 of a gas at STP is filled in a 748 dm3 container. If temperature is constant, calculate the percentage change in pressure required.
Answer:
Initial volume = V1 = 561 dm3
Final volume = V2 = 748 dm3
Difference in volume = 748 – 561 = 187 dm3
As the temperature is constant,
Decrease in pressure percentage =
(187×100)/748 = 25%
Question 9.
88 cm3 of nitrogen is at a pressure of 770 mm mercury. If the pressure is raised to 880 mm Hg, find by how much the volume will diminish, temperature remaining constant.
Answer:
V = 88 cm3
P = 770 mm
P1 = 880 mm
V1= ?
T = T1
Using gas equation,
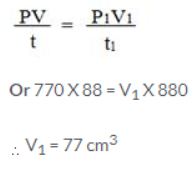
Volume diminishes = 88 – 77 = 11 cm3
Question 10.
A gas at 240 K is heated to 127°C. Find the percentage change in the volume of the gas (pressure remaining constant).
Answer:
Let volume = 100 ml
T = 240 K
Volume increased = x ml
New volume = 100 + x ml
T1 = 400 K
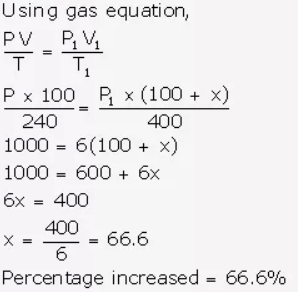
Question 11.
Certain amount of a gas occupies a volume of 0.4 litre at 17°C. To what temperature should it be heated so that its volume gets (a) doubled, (b) reduced to half, pressure remaining constant?
Answer:
(a) V1 = 0.4 L
V2 = 0.4 × 2L
T1 = 17°C (17 + 273) = 290 K
T2= ?
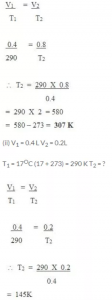
=145 – 273 = -128°C
Question 12.
A given mass of a gas occupies 143 cm³ at 27°C and 700 mm of Hg pressure. What will be its volume at 300 K and 280 mm of Hg pressure?
Answer:
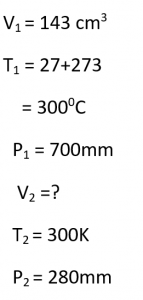
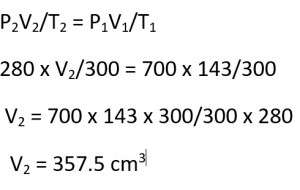
Question 13.
A gas occupies 500 cm3 at normal temperature. At what temperature will the volume of the gas be reduced by 20% of its original volume, pressure being constant?
Answer:
V = 500 cm3
Normal temperature, t = 0°C = 0 + 273 K
V1 = Reduced volume + 20% of 500 cm3

Question 14.
Calculate the final volume of a gas ‘X’ if the original pressure of the gas at STP is doubled and its temperature is increased three times.
Answer:

Question 15.
A sample of carbon dioxide occupies 30 cm3 at 15°C and 740 mm pressure. Find its volume at STP.
Answer:

F. Exercise – 7 Numericals Answer Type
Study of Gas Laws Class-9 Chemistry Concise Solutions
Page-134
Question 16.
What temperature would be necessary to double the volume of a gas initially at STP if the pressure is decreased to 50%?
Answer:
V1 = V1
P1 = 760 atm
T1 = 273 K
V2 = 2V1
T2 =?

Question 17.
At 0°C and 760 mmHg pressure, a gas occupies a volume of 100 cm3. Kelvin temperature of the gas is increased by one-fifth and the pressure is increased one and a half times. Calculate the final volume of the gas.
Answer:

Question 18.
It is found that on heating a gas its volume increases by 50% and its pressure decreases to 60% of its original value. If the original temperature was -15°C, find the temperature to which it was heated.
Answer:
Let the original volume (V) = 1 and
the original pressure (P) = 1 and
the temperature given (T) = -15°C = -15 + 273 = 258 K
V1 or new volume after heating = original volume + 50% of original volume

Question 19.
A certain mass of a gas occupies 2 litres at 27°C and 100 Pa. Find the temperature when volume and pressure become half of their initial values.
Answer:

Question 20.
2500 cm3 of hydrogen is taken at STP. The pressure of this gas is further increased by two and a half times (temperature remaining constant). What volume will hydrogen occupy now?
Answer:

Question 21.
Taking the volume of hydrogen as calculated in Q.19, what change must be made in Kelvin (absolute) temperature to return the volume to 2500 cm3 (pressure remaining constant)?
Answer:

Question 22.
A given amount of gas A is confined in a chamber of constant volume. When the chamber is immersed in a bath of melting ice, the pressure of the gas is 100 cm Hg.
(a) What is the temperature when the pressure is 10 cmHg?
(b) What will be the pressure when the chamberis brought to 100°C
Answer:


Question 23.
A gas is to be filled from a tank of capacity 10,000 litres into cylinders each having capacity of 10 litres. The condition of the gas in the tank is as follows:
(a) Pressure inside the tank is 800 mmHg.
(b) Temperature inside the tank is -3°C.
When the cylinder is filled, the pressure gauge reads 400 mmHg and the temperature is 0°C. Find the number of cylinders required to fill the gas.
Answer:
Capacity of the cylinder V = 10000 litres
P = 800 mm
T = -3°C = -3 + 273 = 270 K P1 = 400 mmHg
T1 = 0°C = 0 + 273 = 273 K
V1= ?

Question 24.
A sealed tube (able to withstand a pressure of 3 atmospheres) is filled with air at 27°C and one atmosphere pressure. Find the temperature above which it will burst.
Answer:
P1 = Initial pressure of the gas = 1 atm
T1 = Initial temperature of the gas = 27°C = 27 + 273 = 300 K
P2 = Final pressure of the gas = 3 atm
T2 = Final temperature of the gas = ?
V1 = V2 = V [constant volume]
By Gas Law:

∴ The temperature above which the tube will burst is 627°C.
Question 25.
Calculate the temperature at which the volume of a gas is doubled if its pressure at the same time increases from 70 cm of Hg to 80 cm of Hg.
Answer:
Initial conditions :
P1 = Initial pressure of the gas = 70 cm of Hg
V1 = Initial volume of the gas = V
T1 = Initial temperature of the gas = 273 K
Final conditions:
P2 (Final pressure) = 80 cm of Hg
V2 (Final volume) = 2V
T2 (Final temperature) = ?
By Gas Law:
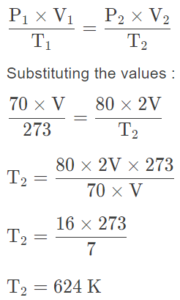
∴ Final temperature of the gas = 624 – 273 = 351°C
Question 26.
Calculate the volume occupied by 2 g of hydrogen at 27°C and 4 atmosphere pressure if at STP it occupies 22.4 litres.
Answer:
V1 = 22.4 litres
P1 = 1 atm
T1 = 273 K
V2 =?
T2 = 300 K
P2 = 4 atm

Question 27.
Which will have greater volume when the following gases are compared at S.T.P.
(a) 1.2 l N2 at 25°C and 748 mm Hg
(b) 1.25 l O2 at S.T.P.?
Answer:
(a) 1.2 l N2 at 25°C and 748 mm Hg
Initial conditions :
P1 = Initial pressure of the gas = 748 mm Hg
V1 = Initial volume of the gas = 1.2 l
T1 = Initial temperature of the gas = 25°C = 25 + 273 = 298 K
Final conditions :
P2 (Final pressure) = 760 mm of Hg
T2 (Final temperature) = 273 K
V2 (Final volume) = ?
By Gas Law:
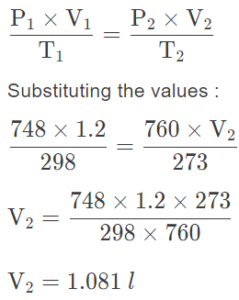
∴ Volume of N2 at S.T.P. = 1.081 l and volume of O2 at S.T.P. is 1.25 l. Hence, volume of O2 at S.T.P. is greater than N2 at S.T.P.
(Study of Gas Laws Exe-7 Numericals Answer Chemistry Class-9)
Question 28.
50 cm3 of hydrogen is collected over water at 17°C and 750 mmHg pressure. Calculate the volume of a dry gas at STP. The water vapour pressure at 17°C is 14 mm Hg.
Answer:
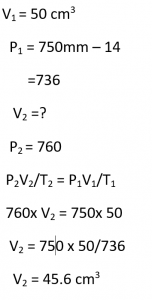
Question 29.
Calculate the volume of dry air at STP that occupies 28 cm3 at 14°C and 750 mmHg pressure when saturated with water vapour. The vapour pressure of water at 14°C is 12 mmHg.
Answer:
Pressure due to dry air,
P = 750 – 12 = 738 mm
V = 28 cm3
T = 14°C = 14 + 273 = 287 K
P1 = 760 mmHg
V1= ?
T1 = 0°C = 273 K
Using gas equation,

Question 30.
LPG cylinder can withstand a pressure of 14.9 atmosphere. The pressure gauge of the cylinder indicates 12 atmosphere at 27°C. Because of a sudden fire in the building, the temperature rises. At what temperature will the cylinder explode?
Answer:
P = 14.9 atm
V = 28 cm3
T = ?
P1 = 12 atm
V = V1
T1 = 300 K
Using gas equation,

Question 31.
22.4 litres of a gas weighs 70 g at STP. Calculate the weight of the gas if it occupies a volume of 20 litres at 27°C and 700 mmHg of pressure.
Answer:
Step 1:
V1 = 20 litres
P1 = 700 mm
T1 = 300 K
V2= ?
T2 = 273 K
P2 = 760 mm


— : End of Study of Gas Laws Exe-7 Numericals Answer Class-9 ICSE Chemistry Solutions :–
Return to Return to Concise Selina ICSE Chemistry Class-9
Thanks
Please share with your friends




