Water Exe-3A Chemistry Class-9 ICSE Selina Publishers Solutions Chapter-3. Step By Step ICSE Selina Concise Solutions of Chapter-3 Water with All Exercise including MCQs, Very Short Answer Type, Short Answer Type, Long Answer Type, Numerical and Structured/Application Questions Solved . Visit official Website CISCE for detail information about ICSE Board Class-9.
Water Exe-3A Chemistry Class-9 ICSE Concise Selina Publishers
| Board | ICSE |
| Publications | Selina Publication |
| Subject | Chemistry |
| Class | 9th |
| Chapter-3 | Water |
| Book Name | Concise |
| Topics | Solution of (Water) Exercise – 3A |
| Academic Session | 2023-2024 |
A. Exercise – 3A
Water Class-9 Chemistry Concise Solutions
Page-39
Question 1.
Water exists in all three states. Discuss.
Answer:
In the Free State, water occurs in the solid, liquid and gaseous states.
(a) Solid state: A large amount of fresh water is found in the form of snow or ice.
(b) Liquid state: Most of the water present in oceans and found in streams, rivers, lakes, ponds and springs on land is water in the liquid state.
(c) Gaseous state: Water vapour present in the air is in the gaseous state. Water vapour condenses in the sky to form clouds. Mist and fog are also examples of water in the gaseous form
Question 2.
Why water is considered a compound?
Answer:
Water is considered a compound because it is made of two elements hydrogen and oxygen combined in the ratio 1:8 by mass.
Mass ratio of elements H2O
H : O, 2 × 1 : 16 × 1 = 1 : 8
(Atomic mass of H = 1, O = 16)
Components of water cannot be separated by physical methods but can be separated by electrolysis of water.
Question 3.
(a) Why does temperature in Mumbai and Chennai not fall as low as it does in Delhi?
(b) Give the properties of water responsible for controlling the temperature of our body.
Answer:
a) The temperature in costal cities like Mumbai and Chennai do not fall as low as in Delhi because these cities are situated in the coastal areas. Due to high specific heat capacity, the presence of a large amount of water is able to modify the climate of the nearby land areas making them warmer in winter and cooler in summer. So, the temperature does not fall as low as it does in Delhi.
b) Our body is almost 65% of water, and it has the property of specific heat. Due to high specific heat capacity, the presence of a large amount of water is able to modify the climate of the body and control the temperature of our body, which is warm in winter and cool in summer.
Question 4.
‘Water is the universal solvent’. Comment.
Answer:
Water dissolves many substances forming an aqueous Solution. It can dissolve solids, liquids and gases. When a solid dissolves in water, the solid is the solute, the water is the solvent and the resultant liquid is the Solution. So, it is said that water is a universal solvent. In other words, water can dissolve nearly every substance.
Question 5.
What causes the violence associated with torrential rain?
Answer:
The sudden release of the latent heat of condensation causes the violence associated with torrential rain.
Question 6.
(a) Which property of water enables it to modify the climate?
(b) Density of water varies with temperature. What are its consequences?
(c) What is the effect of impurities present in the water on the melting point and boiling point of water?
Answer:
(a)Due to the high specific heat capacity, the presence of a large amount of water is able to modify the climate.
(b)The property of anomalous expansion of water enables marine life to exist in the colder regions of the world, because even when water freezes on the top, it is still liquid below the ice layer, as the density of water is greater than that of ice.
(c)The boiling point of water increases due to the presence of dissolved impurities.
The freezing point of water decreases due to the presence of dissolved impurities.
Question 7.
How do fishes and aquatic animals survive when the pond gets covered with thick ice?
Answer:
Even though ponds covered with thick ice, beneath there will be water because of the maximum density level of water which helps the fishes and aquatic animals survive
Question 8.
The properties of water are different from the properties of the elements of which it is formed. Discuss.
Answer:
Water is composed of Hydrogen and Oxygen in the ratio of 2:1. Its properties are distinct from those of its component elements in the following ways:
| Properties | Hydrogen | Oxygen | Water |
|---|---|---|---|
| State at room temperature | Gas | Gas | Liquid |
| Boiling Point | -253°C | -183°C | 100°C |
| Melting point | –259°C | -218.8°C | 0°C |
| Flammability | Explosive | Necessary for combustion | Extinguishes flame |
| Polarity | Non-polar | Non-polar | Polar |
Question 9.
How is aquatic life benefited by the fact that water has maximum density at 4oC?
Answer:
The property of anomalous expansion of water enables aquatic life to exist because water freezes on the surface of the water body, but it is still liquid below the ice layer.
Question 10.
What are the observations and conclusions when tap water is boiled and evaporated in a water glass?
Answer:
When tap water is boiled and evaporated:
Observations:
(a) A number of concentric rings of solid matter are seen on the watch glass after evaporation of tap water.
Conclusion:
(b) Tap water contains dissolved salts, minerals and impurities.
Question 11.
What is the importance of dissolved salts in water?
Answer:
Importance of dissolved salts in water:
(a)Dissolved salts provide specific taste to water.
(b)Dissolved salts act as micronutrients for the growth and development of living beings.
Question 12.
State the importance of the suitability of CO2 and O2 in water.
Answer:
They add taste to water for drinking purposes.
Question 13.
How is air dissolved in water different from ordinary air?
Answer:
Oxygen is more soluble in water than nitrogen. Air dissolved in water contains a higher percentage of oxygen (30-35%). Oxygen is only 21% in ordinary air. In this way, air dissolved in water is different from ordinary air.
Question 14.
Identify A, B, C, and D first one is done for you.
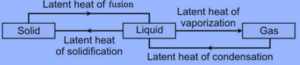





Answer:
When a solid changes into a liquid, it absorbs heat equal to the latent heat of fusion. When a liquid changes into a solid, it loses heat equal to the latent heat of solidification.
a liquid changes into a gas, it absorbs heat equal to the latent heat of vaporization. When a gas condenses into a liquid, it loses heat equal to the latent heat of condensation.
Question 15.
Explain why:
(a) Boiled or distilled water tastes flat.
(b) Ice at zero degrees centigrade has greater cooling effect than water at 0oC.
(c) Burns caused by steam are more severe than burns caused by boiling water.
(d) Rivers and lakes do not freeze easily?
(e) Air dissolved in water contains a higher proportion of oxygen.
(f) If distilled water is kept in a sealed bottle for a long time, it leaves etchings on the surface of the glass.
(g) Rain water does not leave behind concentric rings when boiled.
Answer:
(a) Boiled water tastes flat because it does not contain dissolved matter such as air, carbon dioxide and other minerals.
(b) Ice at 0°C gives more cooling effect than water at 0°C because at 0°C ice absorbs 336 J per gram of energy to melt to 0°C water.
(c) Burns caused by steam are more severe than burns caused by boiling water because of high specific latent heat of condensation. 2268 J/g of heat is released when 1 g of steam condenses to form 1 gm of water.
(d) Due to the high specific latent heat of solidification of water, rivers and lakes do not freeze easily.
(e) Air dissolved in water contains a higher percentage of oxygen because the solubility of oxygen in water is more than that of oxygen in air.
(f)If distilled water is kept in a sealed bottle for a long time, it etches the surface of glass because substances which are apparently insoluble in water actually dissolve in minute traces in water.
(g)Rain water does not leave concentric rings when boiled because rain water does not contain dissolved solids.
— : End of Water Exe-3A Answer Class-9 ICSE Chemistry Solutions :–
Return to Return to Concise Selina ICSE Chemistry Class-9
Thanks
Please share with your friends




