Water Exe-3C Descriptive Answer Chemistry Class-9 ICSE Selina Publishers Solutions Chapter-3. Step By Step ICSE Selina Concise Solutions of Chapter-3 Water with All Exercise including MCQs, Very Short Answer Type, Short Answer Type, Long Answer Type, Numerical and Structured/Application Questions Solved . Visit official Website CISCE for detail information about ICSE Board Class-9.
Water Exe-3C Descriptive Answer Chemistry Class-9 ICSE Concise Selina Publishers
| Board | ICSE |
| Publications | Selina Publication |
| Subject | Chemistry |
| Class | 9th |
| Chapter-3 | Water |
| Book Name | Concise |
| Topics | Solution of (Soft and hard Water) Exercise – 3C Descriptive Answer |
| Academic Session | 2023-2024 |
Exercise – 3C Descriptive Answer Type
Water Class-9 Chemistry Concise Solutions
Page-55
Question 1.
What are hydrous substances? Explain with examples.
Answer :
Substances which contain water molecules along with salt are hydrated substances.
Examples: Sodium carbonate dehydrate: Na2CO3.10H2O
Copper sulphate pentahydrate: CuSO4.5H2O
Question 2.
What is the importance of dissolved impurities in water?
Answer:
The dissolved impurities in water are salts and minerals.
Dissolved salts provide specific taste to water.
Salts and minerals are essential for growth and development.
They supply the essential minerals needed by our body.
Question 3.
What are the advantages of:
(i) soft water
(ii) hard water?
Answer:
Advantages of soft water:
(1) When the water is soft, you use much less soap and fewer cleaning products. Your budget will reflect your savings.
(2)Plumbing will last longer. Soft water is low in mineral content and therefore does not leave deposits in the pipes.
(3)Clothes last longer and remain bright longer if they are washed in soft water.
Advantages of hard water:
(1) Water free from dissolved salts has a very flat taste. The presence of salts in hard water makes it tasty. So, hard water is used in making beverages and wines.
(2) Calcium and magnesium salts present in small amounts in hard water are essential for bone and teeth development.
(3) Hard water checks the poisoning of water by lead pipes. When these pipes are used for carrying water, some lead salts dissolve in water to make it poisonous. Calcium sulphate present in hard water forms insoluble lead sulphate in the form of a layer inside the lead pipe and this checks lead poisoning.
Question 4.
What are stalagmites and stalactites? How are they formed?
Answer:
In some limestone caves, conical pillar-like objects hang from the roof and some rise from the floor. These are formed by water containing dissolved calcium hydrogen carbonate continuously dropping from the cracks in the rocks. Release of pressure results in the conversion of some hydrogen carbonate to calcium carbonate.
Ca (HCO3)2 → CaCO3 + CO2 + H2O
This calcium carbonate little by little and slowly deposit on both roof and floor of the cave.
The conical pillar which grows downwards from the roof is called stalactite and the one which grows upward from the floor of the cave is called stalagmite.
These meet after a time. In a year, some grow less than even a centimetre, but some may be as tall as 100 cm.
CaCO3 + CO2 + H2O → Ca (HCO3)2
MgCO3 + CO2 + H2O → Mg (HCO3)2
If the water flows over beds of gypsum (CaSO4.2H2O), a little bit of gypsum gets dissolved in water and makes it hard.
Question 5.
Give equations to show what happens when temporary hard water is
(a) Boiled
(b) Treated with slaked lime
Answer:
(a) when boiled
Ca (HCO3)2 → CaCO3 + H2O + CO2↑
Mg (HCO3)2 → MgCO3 + H2O + CO2↑
(b) when Treated with slaked lime
Ca(HCO3)2+ Ca(OH)2 → 2CaCO3 + 2H2O
Mg (HCO3)2+ Ca(OH)2→ MgCO3 + 2H2O
Question 6.
State the disadvantages of using hard water.
Answer:
It is more difficult to form lather with soap.
Scum may form in a reaction with soap, wasting the soap.
Carbonates of calcium and magnesium form inside kettles. This wastes energy whenever you boil a kettle.
Hot water pipes ‘fur up’. Carbonates of calcium and magnesium start to coat the inside of pipes which can eventually get blocked.
Question 7.
Why does the hardness of water render it unfit for use in a
(i) boiler
(ii) for washing purposes.
Answer:
Steam is usually made in boilers which are made of a number of narrow copper tubes surrounded by fire. As the cold water enters these tubes, it is immediately changed into steam, while the dissolved solids incapable of changing into vapour deposit on the inner walls of the tubes.
This goes on and makes the bore of the tubes narrower. The result is that less water flows through the tubes at one time and less steam is produced. When the bore of the tube becomes very narrow, the pressure of the steam increases so much that at times the boiler bursts.
If hard water is used,
calcium and magnesium ions of the water combine with the negative ions of the soap to form a slimy precipitate of insoluble calcium and magnesium usually called soap curd (scum).
Formation of soap curd will go on as long as calcium and magnesium ions are present. Till then, no soap lather will be formed and cleaning of clothes or body will not be possible. Moreover, these precipitates are difficult to wash from fabrics and sometimes form rusty spots if iron salts are present in water.
Question 8.
Explain with equation, what is noticed when permanent hard water is treated with
(a) Slaked time
(b) Washing soda
Answer :
(a) Slaked lime
Ca (HCO3)2 + Ca(OH)2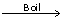
ICSEHELP Premium Slider
✨ SPECIAL NOTES DOWNLOAD NOW ✨
 10TH CHEMISTRY
DOWNLOAD NOW
10TH CHEMISTRY
DOWNLOAD NOW
 10TH MATH
DOWNLOAD NOW
10TH MATH
DOWNLOAD NOW
 10TH PHYSICS
DOWNLOAD NOW
10TH PHYSICS
DOWNLOAD NOW
 10TH BIOLOGY
DOWNLOAD NOW
10TH BIOLOGY
DOWNLOAD NOW
 12TH BIOLOGY
DOWNLOAD NOW
2CaCO3 + 2H2O
12TH BIOLOGY
DOWNLOAD NOW
2CaCO3 + 2H2O





Mg (HCO3)2+ Ca(OH)2 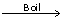 MgCO3 + CaCO3 + 2H2O
MgCO3 + CaCO3 + 2H2O
Lime is first thoroughly mixed with water in a tank and then fed into another tank containing hard water. Revolving paddles thoroughly mix the two Solutions. Most of the calcium carbonate settles down. If there is any solid left over, it is removed by a filter. This is known as Clarke’s process.
(b) Washing soda
When washing soda or soda ash is added to hard water, the corresponding insoluble carbonates settle down and can be removed by filtration.
Ca (HCO3)2 + Na2CO3 —–> CaCO3 + 2NaHCO3
Mg (HCO3)2+ Na2CO3 —–> MgCO3 + 2NaHCO3
Question 9.
What is permutit method, how can it be used for softening hard water ?
Answer:
— : End of Water Exe-3C Descriptive Answer Class-9 ICSE Chemistry Solutions :–
Return to Return to Concise Selina ICSE Chemistry Class-9
Thanks
Please share with your friends




